| ** | Latin American Journal of Clinical Sciences and Medical Technology is an open access magazine. To read all published articles and materials you just need to register Registration is free of charge. Register now If you already have registered please Log In | ** |
aHematológica, Alta Especialidad, Hospital Ángeles Lomas, Estado de México, México; bISSSTE Hospital Regional Tipo B de Alta Especialidad Bicentenario de la Independencia; cIMSS Hospital General Regional No. 1 “Dr. Carlos Mac Gregor Sánchez Navarro”.
Corresponding Author: , . Telephone number: ; e-mail: drabaez.hematologia@gmail.com
Lat Am J Clin Sci Med Technol. 2023 Oct;5:258-267.
Received: May 18th, 2023.
Accepted: September 22nd, 2023.
Published: October 18th, 2023.
Views: 1667
Downloads: 9
Background. The COVID-19 disease is characterized by its severe form in which pneumonia and cytokine hypersecretion are observed. Currently, one of the main lines of treatment has been aimed at controlling the hyperinflammatory state of the host. Material and Methods. An open-label, multicenter, non-randomized, phase II clinical trial was conducted in patients with moderate to severe SARS-CoV-2 pneumonia treated with ruxolitinib 5 mg twice daily for 14 days along with the standard of care. The use of dexamethasone was allowed under the investigator's discretion. The primary endpoint was respiratory improvement based on the 8-point ordinal scale for severity. Results. Seventy-seven cases were studied in three centers; 62% were men with a mean age of 51±12 years. The most frequent associated comorbidity was overweight. At the beginning of the study, the mean PaO2/FiO2 index was 195 ± 90.9 with mean SatO2 86%; 63 patients had 5 points on the 8-ordinal scale, and 14 World Health Organization. WHO Coronavirus (COVID-19) Dashboard. 2021 [Cited 2021 Jul 16]. Available from URL: https://covid19.who.inthad 6 points. At the end of the study, 58 patients met the primary endpoint of improvement observed in the reduction of a mean 8-point score with a baseline of 5.18 ± 3.8 points and a 15-day score of 2.77 ± 2.63. PaO2/FiO2 showed a constant increase with a final mean of 315.36 ± 76.76. Nine patients died due to critical COVID-19. Thrombocytosis, followed by elevated liver enzymes grade 1, was the most observed adverse event. Conclusions. Ruxolitinib has an adequate profile to treat patients with COVID-19 pneumonia. This study's limitations were the lack of a control group and the small sample size. Future studies must follow the results presented here to corroborate the effectiveness of ruxolitinib.
Antecedentes. La enfermedad COVID-19 se ha caracterizado por su forma severa, manifestada por neumonía e hipersecreción de citocinas. Actualmente, una de las principales líneas de tratamiento ha sido buscar el control del estado hiperinflamatorio del huésped. Material y métodos. Se realizó un estudio abierto, multicéntrico, no aleatorizado, fase II en pacientes con neumonía por SARS-CoV-2 moderada a severa, tratados con ruxolitinib 5 mg cada 12 horas por 14 días junto con el tratamiento estándar. El uso de dexametasona fue permitido bajo el criterio del investigador. El objetivo primario fue la mejoría respiratoria medida por la escala ordinal de 8 puntos para severidad. Resultados. Se estudiaron setenta y siete casos en tres centros, 62% fueron hombres, con una edad promedio de 51±12 años. La comorbilidad más frecuentemente asociada fue el sobrepeso. Al inicio del estudio, la media de PaO2/FiO2 fue 195 ± 90.9 y SatO2 de 86%; 63 pacientes tenían 5 puntos en la escala de 8 puntos y 14 tenían 6 puntos. Al final del estudio 58 pacientes cumplieron el objetivo primario de mejoría observado con reducción de la escala de 8 puntos con un basal de 5.18 ± 3.8 puntos y un puntaje a 15 días de 2.77 ± 2.63. El índice de PaO2/FiO2 mostró aumento constante con una media final de 315.36 ± 76.76. Nueve pacientes fallecieron por COVID-19 severo. El evento adverso más observado fue trombocitosis, seguido por elevación de enzimas hepáticas grado 1. Conclusiones. Ruxolitinib mostró un perfil adecuado para su uso en pacientes con neumonía por COVID-19. Las limitaciones del estudio fueron que carecía de un grupo de control y que la muestra era pequeña. Los resultados de ese estudio deben corroborarse con otros acerca de la efectividad de ruxolitinib.
It has been over two years since the World Health Organization (WHO) called the SARS-CoV-2 a pandemic, with a total of 188,655,968 confirmed cases and 4,067,517 deaths in the following eighteen months, since the description of the virus.1,2 On May 5, 2023, WHO terminated the health emergency due to SARS-CoV-2; however, severe viral symptoms with respiratory involvement continue.
So far, the only drug approved by the Food and Drug Administration (FDA) and European Medicines Agency (EMA) is remdesivir. Emergency authorization for monoclonal antibodies and immunomodulators has limited options.3,4
Even with the massive vaccination campaign, there are still cases of the population at risk from severe COVID-19, which is greater among people with limited access to vaccines or new therapies. That is the aim of researching effective and accessible drugs to treat patients with severe COVID-19.
When SARS-CoV-2 adheres to the alveolar epithelium, there is an activation of innate and adaptative immune responses with induction of inflammatory response and subsequent release of cytokines, being the most notable interleukins (IL6, IL-7, IL-10, IL-1β, IL-13), tumour necrosis factor-alpha (TNF-α), and colony-stimulating factors in a dysregulated and exaggerated manner in form of cytokine release syndrome.2,5,6
This hyperactivation of the immune system has motivated diverse clinical trials with immunomodulators to treat severe COVID-19, including the ACTT-2, STOP-COVID, and the COV-BARRIER trials, where Janus tyrosine kinase (JAK) inhibitors were used with favourable responses. With the inhibition of the JAK pathway, the activation of nuclear translocations of Signal Translator and Transcription Activator (STAT) is blocked, modulating the production and secretion of interferon and IL-6 implicated in the pathogenesis of the cytokine release syndrome.7-10 Here, we present the results of ruxolitinib, a JAK 1/2 inhibitor, in an investigator initiative phase II clinical trial.
Study Design
An open-label, prospective, multicenter, single-group, phase II trial was conducted. There were three recruitment centers, all in the metropolitan area of Mexico City. The clinical trial was registered in clinicaltrials.gov as NCT04334044 (Trial registration: Protocol ID HAL 345/2020. Clinicaltrials.gov NCT04334044. Registered 3 April 2020, https://clinicaltrials.gov/ct2/show/NCT04334044 COFEPRIS: CINC42A0MX01T Version 1.2.)
Endpoints and Objectives
The main objective was to measure efficacy in treating pulmonary damage due to SARS-CoV-2 pneumonia. The primary endpoint had a response at 15 days of treatment, defined as a decrease or cessation of oxygen supplementation measured by FiO2 required to hold SpO2 >90%.
The final responses were labeled as clinical response in hospitalized patients, clinical response with hospital discharge, failure to treatment, death, or cessation due to treatment toxicity.
The study used the 8-point ordinal scale developed by the National Institutes of Health (NIH) for the ACTT1 trial to compare patient status at baseline and end of treatment to measure the clinical response. Such scale categorizes the patients as:
- Not hospitalized and without limitation on activities;
- Not hospitalized with limitation on activities and/or requiring home oxygen;
- Hospitalized, not requiring supplemental oxygen, and without ongoing medical care;
- Hospitalized, not requiring supplemental oxygen, with ongoing medical care;
- Hospitalized, requiring supplemental oxygen;
- Hospitalized, using high-flow non-invasive oxygen devices;
- Hospitalized, on invasive mechanical ventilation or extracorporeal membrane oxygenation;
- Dead.
Secondary objectives were the length of intrahospital stay; requirement of mechanical ventilation (MV); death due to COVID-19; changes in inflammatory laboratory values measured with C-reactive protein (CRP), lactic dehydrogenase (LDH), erythrocyte sedimentation rate (ESR) and ferritin levels; and toxicity to ruxolitinib.
Selection Criteria
Inclusion criteria comprise 18-year-old patients (or older), diagnosed with COVID-19 from a confirmatory test of SARS-CoV-2 and evidence of pulmonary damage by:
- Dyspnea or increased breathing work demonstrated by a respiratory rate of ≥20 bpm.
- Pulse oxygen saturation <90% with FiO2 21% or PaO2 <65 mmHg in case of oxygen supplementation.
- Pulmonary changes on radiographic images (X-ray or CT) compatible with COVID-19.
Exclusion criteria included pregnancy or breastfeeding; chronic renal failure with creatinine clearance <15 mL/min; Charlson comorbidity index ≥5 points; thrombocytopenia <20,000 cells/mm3; neutropenia <500 cells/mm3; known active infection of human immunodeficiency virus, hepatitis C, hepatitis B, herpes zoster or tuberculosis; and concomitant use of tocilizumab, baricitinib or interferon. Elimination criteria were voluntary withdrawal of informed consent and grade ≥3 adverse events to ruxolitinib.
Study Intervention and Follow-up
All the selected patients were treated with ruxolitinib 5 mg twice daily for 15 days. The trial drug was suspended in case of treatment failure, defined as pneumonia deterioration with required mechanical ventilation or death.
Ruxolitinib was granted with the standard of care of each research center according to the physician’s criteria. Novartis Farmacéutica S.A de C.V. provided it during the entire trial with protocol number CINC424A0MX01T. No other financial support was given. Clinical status was analyzed daily with the registration of an 8-point ordinal scale and laboratory values on days 1, 5, 10, and 15 of treatment. All severe adverse events were informed to the Ethical Local Committee, National Health Authority (COFEPRIS), and Novartis pharmacovigilance program.
Ethical Issues and Compliance
Participation in this study was voluntary in all cases, certified by an informed consent process. The study was developed and carried out following current national and international regulations. It was submitted for approval by Hospital Ángeles Investigation and Ethics Committee as the Federal Commission for the Protection against Sanitary Risk. Novartis was neither involved in the collection nor the data analysis.
Statistical Analysis
The leading investigator assembled the registration of the patients in Office Excel spreadsheets. The statistical analysis was elaborated in the IBM SPSS Statistics v.22.0 program. Descriptive analysis was carried out with measures of central tendency and dispersion. The comparatives between categorical variables were performed with a Chi-square test as ANOVA for quantitative variables. For non-parametric variables, a Mann-Whitney U test was conducted. The measurements of survival were performed with Kaplan-Meier curves. We considered statistically significant a value of p <0.05.
Patient Characteristics
Seventy-seven patients were included in the study from August 11th 2020 to April 12th 2021 in three medical centers in Mexico City.
Three patients voluntarily withdrew their consent, two of them due to hospital discharge. Seventy-seven patients were analyzed for safety, and 74 were studied for efficacy (Table 1).
| Tabla 1. Characteristics of patients at baseline | ||
|---|---|---|
Sex | No. | (%) |
| Female | 29 | (37.7) |
| Male | 48 | (62.3) |
Age - mean (SD) | 52.5 | (11.7) |
| Previous coexisting comorbidities | ||
| Type 2 diabetes | 24 | (31.2) |
| Hypertension | 23 | (29.9) |
| Chronic lung disease | 1 | 1 (1.3) |
| Smoking | 13 | (16.9) |
| Overweight (BMI ≥25) | 62 | (80.5) |
| Obesity (BMI ≥30) | 26 | (33.8) |
Body Mass Index (SD) | 29.6 | (5.4) |
| Concomitant treatment | ||
| Enoxaparin | 66 | (85.7) |
| Apixaban | 11 | (14.3) |
| Systemic corticosteroids | 63 | (81.8) |
| Azithromycin | 31 | (40.3) |
| Doxycycline | 28 | (36.4) |
| Ceftriaxone | 7 | (9.1) |
| Clarithromycin | 2 | (2.6) |
| Meropenem | 2 | (2.6) |
| Other antibiotics | 3 | (3.9) |
Oxygen saturation -mean (SD) PaFiO2 | 86.5 195.1 | (8.1) (14.3) |
| Supplemental oxygen | 77 | (100) |
| Nasal cannula | 12 | (15.6) |
| Face mask | 6 | (7.8) |
| High-flow nasal cannula | 47 | (61.0) |
| High-flow face mask | 12 | (15.6) |
| 8-point ordinal scale | ||
| 5 points | 63 | (81.8) |
| 6 points | 14 | (18.2) |
| SD: standard deviation; BMI: body mask index; PaFiO2: partial pressure arterial oxygen and fraction of inspired oxygen ratio.The table is property of the authors. | ||
Most patients were male (62.3%) with a mean age of 52±12 years. A history of type 2 diabetes was present in 31.2%, the most frequent comorbidity followed by hypertension in 29.9% and smoking in 16.9%. Two patients had chronic lung diseases; two patients had chronic kidney disease; there were two patients with hypothyroidism; one case of rheumatoid arthritis, and one subject with type 1 diabetes.
There was a tendency towards overweight, with a body mass index (BMI) greater than 25 in 80.5% of the population. BMI classified the cases as overweight in 36 cases and obesity in 22 cases, and severe obesity in 4 cases. When comorbidities and health status were classified according to the Charlson comorbidity index, 52 patients had 0 points, 20 patients had 1 point, 2 had 4 points, and one patient had 3 points.
All the patients required oxygen support with a peripheral oxygen saturation of 86.5% ± 8.1% and a Kirby index (Pa/FiO2) of 195.1 ± 90.9. Oxygen supplementation was given with a conventional nasal cannula or face mask in 59 patients, while 18 had high-flow oxygen devices. According to the 8-point ordinal scale, 81.8% of the cases had 5 points, and 18.2% had 6 points. There were no patients on invasive mechanical ventilation nor extracorporeal membrane oxygenation; therefore, there were no patients with 7 points in the ordinal scale at baseline.
Sixty-seven patients were treated with antibiotics; 31 cases used azithromycin, 28 doxycycline, 7 ceftriaxone, 2 clarithromycin, 2 meropenem, 1 amoxicillin, 1 cefepime, and one levofloxacin.
All the patients had anticoagulant treatment, with the most frequently used enoxaparin (66 cases); the rest were treated with apixaban. Sixty-three patients were on systemic corticosteroids; 58 had dexamethasone, and 5 had methylprednisolone. Only three cases were treated with atorvastatin, and two patients had ivermectin in their treatments. The mean days from diagnosis to treatment with ruxolitinib was 3 days±2.82 ranging from 1 to 5 days.
Primary Endpoint
The primary efficacy objective was measured by weaning or total suspension of oxygen support; 74 patients were evaluated.
The percentage of patients who showed improvement with ruxolitinib treatment was 78.4% (Figure 1). The rest were categorized as treatment failure, with 18.9% of the remaining population showing progression of COVID-19 pneumonia measured by increased oxygen support or death.Two patients remained hospitalized (out of the 58 patients that showed benefit from treatment), while the rest were discharged; put of this group of patients, 13 remained on oxygen at home.
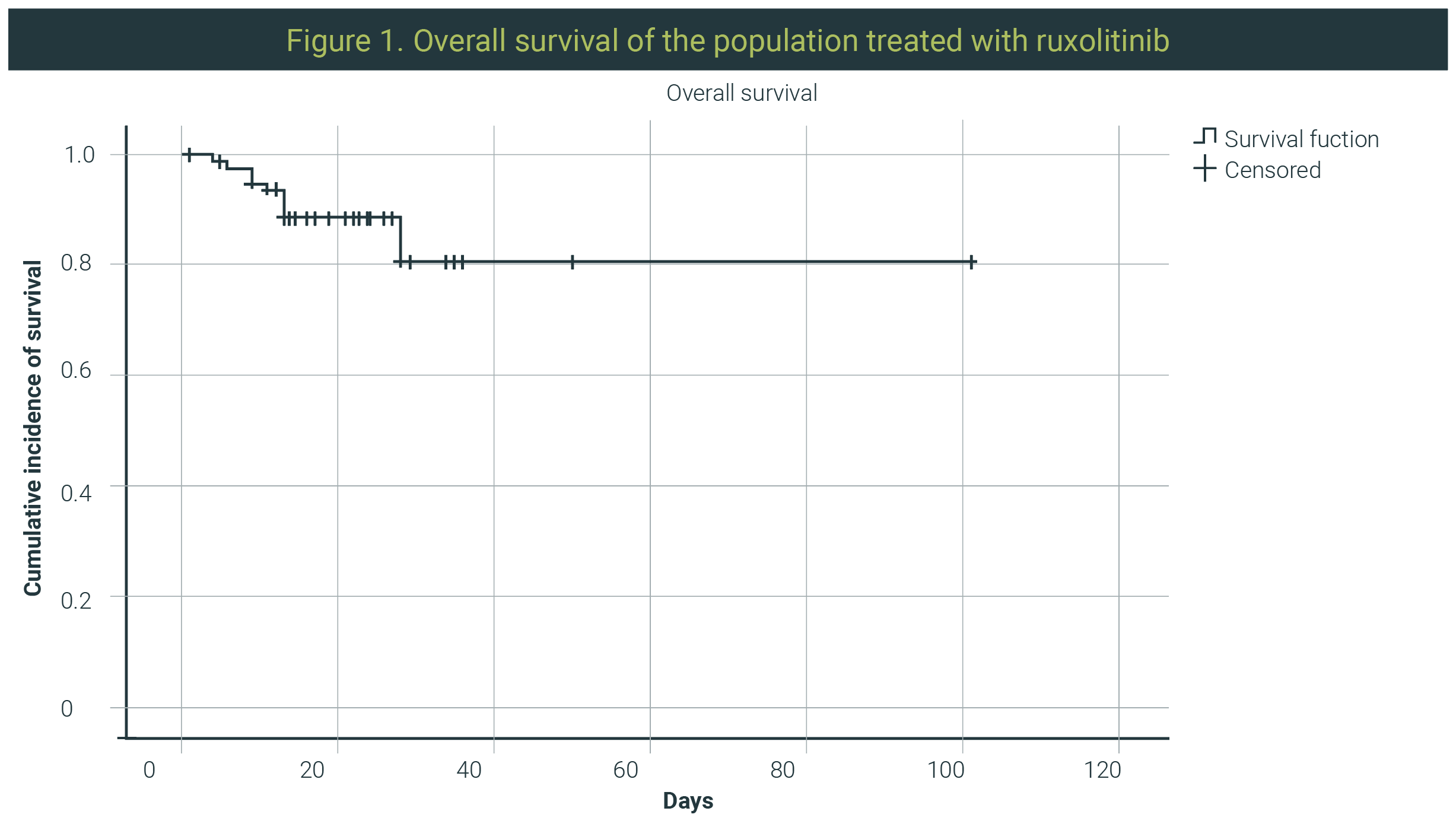
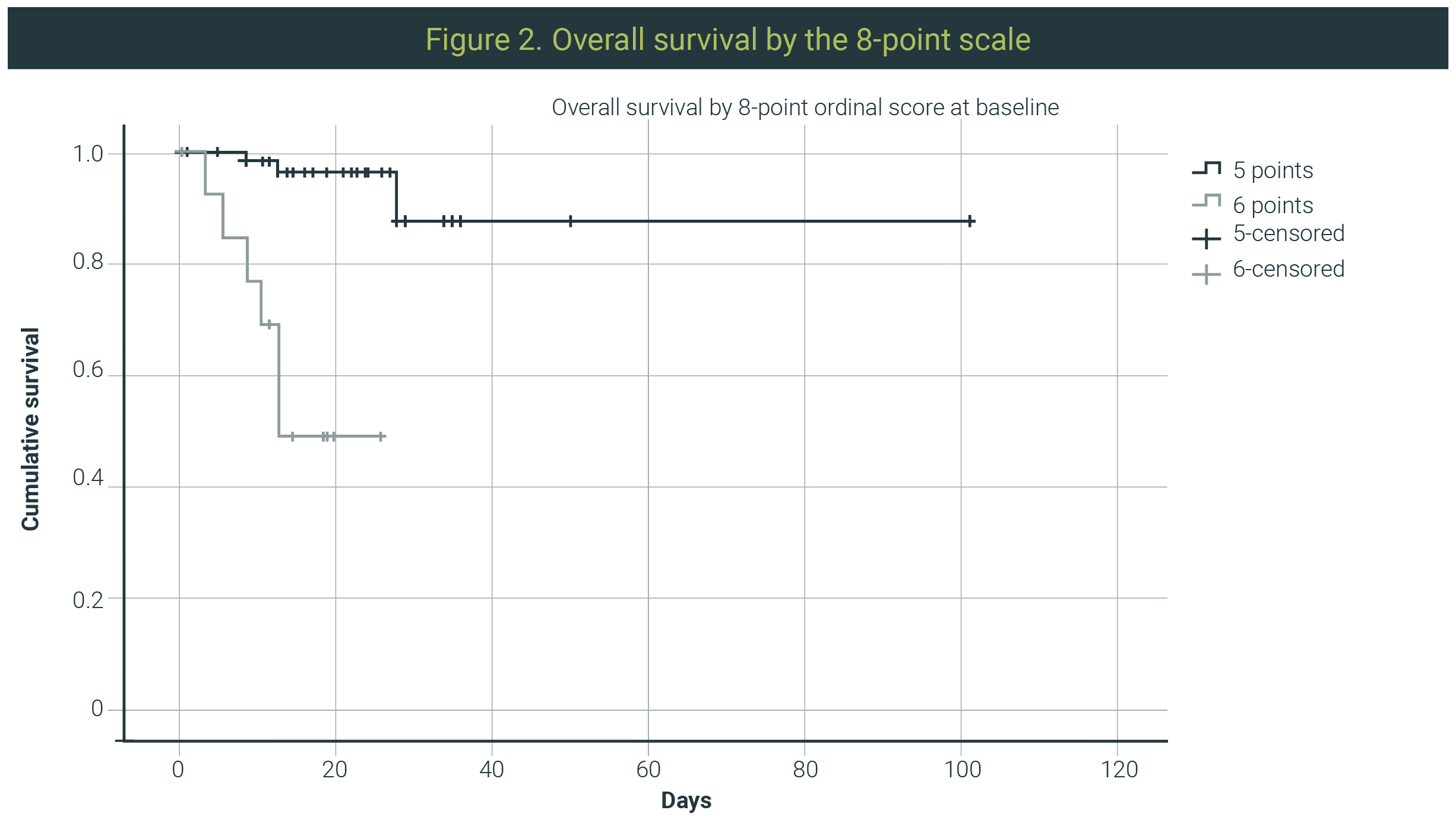
On the one hand, on evaluation by the 8-point ordinal scale measured as a category for each point (as the scale is intended), 42 patients had 1 point score, 14 had 2 points qualifying these patients as improved with the addition of 2 more patients that started with 6 points and ended with 5 points. On the other, qualified as failure to treatment, 2 patients remained with 5 points from start to end of the study; 5 patients ended with 7 points, and 9 deaths scored 8 points (Figure 2).
When efficacy was measured by central tendency measures of the 8-point ordinal scale, a 2.4 reduction in mean points was found. The average of the initial 8-point scale was 5.18±3.8 versus 2.77±2.63 for the final 8-point scale.
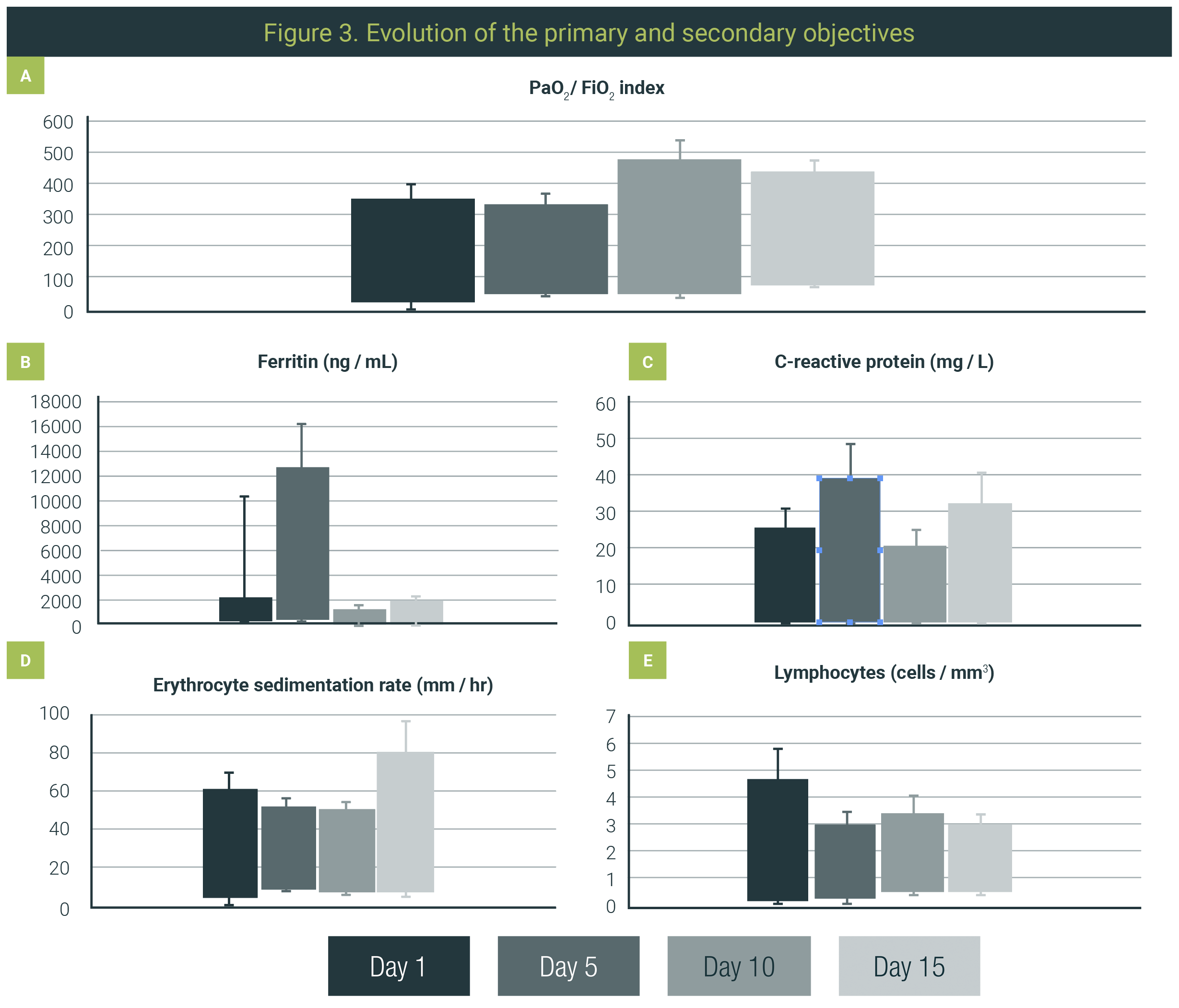
Pa/FiO2 ratio was another measure for oxygenation changes, which had an initial mean of 195.14 ± 90.93 with a steady increase to 222.01±93.94 at day 5 of treatment, 272.45±105.12 at day 10, and 315.36±76.76 after 15 days of treatment.
Secondary Objectives
Sixty-three patients achieved hospital discharge (81.8%) with an in-hospital length of stay of 10.7 ± 6.16 days after beginning treatment. The total hospital stay for the patients was 13.35 ± 7.32 days.
Fourteen (18.2%) patients, out of all the treated patients, progressed to assisted mechanical ventilation, and 9 (11.7%) died. The cause of death in the 9 patients was respiratory failure due to COVID-19 pneumonia.
While searching for prognostic parameters, we analyzed overweight, obesity, sex, diabetes, hypertension, initial serum ferritin levels, initial serum C-reactive protein levels, and baseline Pa/FiO2 index, the latter being the only one that showed statistical significance (p 0.001). That was consistent with higher mortality in those patients who started with higher oxygen supplementation devices. In the univariate analysis, the only statistically significant variable was having an 8-point score >5 points at baseline (p 0.001). (Figure 2)
Biomarkers
There was a decrease in acute-phase reactants. When CRP was measured, a significant decrease was observed with a mean of 9.05±7.72 mg/L at baseline and 2.39 ± 7.14 mg/L after 15 days of treatment.
LDH also decreased with an initial value of 340.34±131.56 U/L with a final value of 225.11±91.83 U/L.
Ferritin had a mean reduction of 267 ng/mL with an initial value of 850±496.7 ng/mL and a final value of 582.13±468.05 ng/mL.
ESR also reduced with a baseline mean of 33.94±13.11 mm/hr and a final mean of 31.27±14.55 mm/hr (Figure 3).
Safety
During the study, there were 50 adverse events registered in 41 patients, including the nine deaths due to COVID-19 pneumonia; forty of them were grade 1, described by Common Terminology Criteria for Adverse Events.
The most frequent event reported was thrombocytosis without subsequent thrombosis or bleeding in 22 patients; those events were classified as grade 1 (asymptomatic); no intervention was needed. The second most frequent adverse event was alanine transaminase elevation in 7 cases and later alkaline phosphatase elevation and lactate dehydrogenase elevation. There was only one case of hyperglycemia and one case of acneiform dermatitis.
There was also one case of grade 1 lymphopenia; nevertheless, the lymphocyte count tended to increase from lymphopenia, with a mean of 1.11 (0.7 cells/mm3) to a final value of 1.7 (0.75 cells/mm3).
This work presents the results of a population with moderate to severe COVID-19 with the criterion of requiring hospitalization and oxygen supplementation, treated with ruxolitinib as an immunomodulator.
In our study, the main objective of pulmonary improvement was met in 78.4% of the patients, achieving hospital discharge in 96%. When the 8-point score assessed clinical improvement, a total average reduction of 2.4 points from the baseline score to the measurement made at the end of treatment was observed. In addition to the clinical improvement, a decrease in serum markers of inflammation was observed, mainly in CRP, LDH, and ferritin.
Such findings are consistent with previously published studies using JAK inhibitors for treating the cytokine storm in COVID-19.
The first study that published this type of results was the work by Fabrizio Cantini et al., where clinical and laboratory parameter improvement was documented, as well as fewer hospitalization days and less progression to intensive therapy requirement in the baricitinib group compared to controls.11
Later, the ACT-II study corroborated the effectiveness of baricitinib in combination with remdesivir, achieving a decrease in mortality, compared to placebo plus remdesivir except for the group of intubated patients, where it did not show a statistically significant difference.9-12
We currently have the results of the COV-BARRIER study, which compared baricitinib plus dexamethasone, the latter being the most widely used standard of treatment in patients with COVID-19 pneumonia. This study corroborated the efficacy of baricitinib in reducing 30- and 60-day mortality with a lower rate of adverse events than the placebo arm.9-12
At the same time, the effectiveness of ruxolitinib in COVID-19 has been controversial. In December 2020, the phase III RUXCOVID study comparing ruxolitinib + SOC versus placebo + SOC was interrupted as it did not meet the primary endpoint of avoiding progression to mechanical ventilation, intensive care, or death at day 29 of evolution.13 In May 2022, the study was published, concluding that ruxolitinib 5 mg twice daily showed no benefit in the overall study population.14
However, other investigator-initiated studies have demonstrated the effectiveness of ruxolitinib in international studies with decreased mortality compared to the standard of care at each site.15-19
In contrast, other single-arm studies documented maximum mortality of 1/14 and 2/34 cases. The benefit was also observed in the improvement of Pa/FiO2, decrease in hospital stay, decrease in cytokines, and recovery of lymphopenia.15-19
Our working group has also conducted two studies with ruxolitinib in moderate to severe pneumonia. The first was a case-control study in which improvement was observed in the population treated with ruxolitinib with no deaths compared to three deaths in the control group and fewer days of hospital stay.20
Such an outcome prompted us to conduct a prospective observational study of patients with ruxolitinib used under the extended-use program. In this study, 287 patients were enrolled, and clinical and laboratory improvement was detected, except for the group of patients who started treatment under assisted mechanical ventilation, where 74% of this population died.20
Thus, we can observe that in the context of phase II and III clinical trials, the results tend to be better with stricter control of the subjects, also speaking of more optimal results in the early initiation of immunomodulation maneuvers, since in the observational study the most significant number of deaths were in patients who already had severe pneumonia requiring invasive ventilation.
Although with fewer studies, there is also evidence of the effectiveness of tofacitinib, another JAK inhibitor. In the multicenter comparative study against placebo, tofacitinib showed a lower mortality rate of 2.8% with tofacitinib versus 5.5% with placebo, and lower hospitalization rates were also observed, with 84% of patients in the intervention arm discharged from the hospital on day 14 of treatment versus 76% in the control arm.8
In our study, we can corroborate the favorable responses with clinical improvement measured by oxygenation parameters and the 8-point score; however, the study was not free of mortality due to COVID-19. One of the factors that could affect, compared to the studies with baricitinib or tofacitinib, is dosing since the minimum approved dose for ruxolitinib was used (5 mg), while in the ACT-II and COV-BARRIER studies the dose used for baricitinib was 4 mg and in the STOP-COVID study the dose of tofacitinib was 10 mg; these doses are the maximum approved dose for both drugs.8-10
Something to reconsider would be to conduct a phase III clinical trial with ruxolitinib at higher doses (at baseline or in escalation). It should be considered that the maximum approved dose is 20 mg to assess the effectiveness associated with the dose, given that the safety range of this drug for the treatment of COVID-19 has already been proven.21-23
It would be ideal for comparing the use of ruxolitinib together with dexamethasone as the current standard of care since, in our study, a mortality of 11% was observed, which is lower than the 22.9% reported in the RECOVERY study and much better than those patients randomized to the arm without corticosteroid, reported at 25.7% (CI 0.75 to 0.93; P<0.001).24
The variables detected as worse prognostic factors were the initial oxygen status of the patients, which has also been observed with baricitinib using the same 8-point score.9
In our study, univariate analysis of overweight, obesity, diabetes, or hypertension for mortality was performed without detecting a statistically significant association as a prognostic factor for fatal outcomes. That is controversial because some studies have described that being overweight has worse results. Still, at the same time, some studies indicate that only a BMI >35 or even >40 impacts morbidity and mortality, with less relevance given to overweight or mild to moderate obesity.25-27
In our study, the adverse events presented were mild in all their occurrences. Although there were deaths, the investigators assigned these as being due to COVID-19 infection and not directly related to the drug under study.
Despite being an immunomodulator, no changes in lymphocytes were observed; on the contrary, a gradual increase in lymphocytes was documented, from an initial lymphopenia of 1.11 lymphocytes/mm3 to a normal final count, findings observed in all the studies with JAK inhibitors.9,10,17,19,28,29
Regarding the hematological aspect, thrombocytosis was observed, but this was asymptomatic and related to adequate drug response. This was also documented in the study by Iabstrener et al., where thrombocytosis was recorded as a frequent event associated with favorable clinical responses.17
More than 24 months after the COVID-19 pandemic, and despite the extensive vaccination campaign implemented, the fourth wave of infections has become present, and the prevalence of patients hospitalized for severe COVID-19 continues to rise with the description of new, more contagious, and potentially more lethal variants.
Thus, the search for useful drugs for managing COVID-19 complications continues to be of great relevance. Currently, the only drug approved by the FDA and the European Union is remdesivir, with limited emergency approvals for monoclonal antibodies against SARS-COV-2, such as bamlanivimab plus etesevimab, casirivimab-imdevimab, and sotrovimab, which have limited accessibility in the real world.30-32
The FDA authorized molnupiravir as an alternative therapy in nonhospitalized patients with mild to moderate COVID-19 who are at high risk of disease progression when ritonavir-boosted nirmatrelvir (paxlovid) and remdesivir are not available to use.33,34
This authorization was given due to the MOVe-OUT trial, where high-risk, unvaccinated patients were enrolled, showing a reduction in death or hospitalization by 31% versus placebo. Nevertheless, these results were not replicated in a population after three doses of a COVID-19 vaccine in the PANORAMIC trial.33
In May 2023, the FDA approved the use of paxlovid (ritonavir-boosted nirmatrelvir) for the treatment of mild to moderate COVID-19 after the results of the EPIC-HR, where this drug showed a reduction in hospitalization rates or death of an 89% versus placebo. However, this drug is only available from Emergency Use Authorization (EUA) supplies in the United States. This scenario motivates the search for easily available and simple-to-administer options.
Although the SARS-COV-2 health emergency has already ended, infections by this virus will continue, as will the processes of cytokine release syndrome in viral pneumonia. Hence, it is relevant to know these types of outcomes. We also consider it pertinent to publish the work carried out in our country within the framework of the COVID-19 pandemic.
Our study with ruxolitinib supports JAK inhibitors along with dexamethasone as an accessible treatment option for the management of moderate to severe pneumonia and cytokine storm in COVID-19 with acceptable effectiveness and safety profile.
The significant limitations of this study are the sample number and the absence of a control arm. However, its evidence is considered helpful in a real-world study in a developing country where resource optimization is the common denominator. In addition, we believe that this information can be transferred to other conditions with cytokine storm as etiopathogenesis.
We want to thank Dr. Joaquín Espinosa, Dr. Marcelo Iabstrebner, and Dr. Alejandro Madrigal for their comments during this study's development.
Ethics Approval and Consent to Participate
The study was developed and carried out according to the current national and international regulations; Hospital Ángeles Investigation and Ethics Committee approved it. All subjects involved in the study signed an informed consent before inclusion in the trial. Participation in this study was voluntary in all cases.
Consent for Publication
Not applicable
Availability of Data and Materials
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Competing Interests
This study was supported by Novartis providing the study drug for all included patients. No other financial support was received. The authors declare that they have no other conflict of interest to declare.
Novartis provided the drug administered to patients, ruxolitinib, under the Novartis “managed access” program. There was no further economic or other kind of financial support provided by Novartis or any other sponsor.
Authors’ Contributions
Dr. ROM presented the original idea; Dr. XCR, Lic. CCT, and Dr. PEBI were responsible for drafting the protocol and submitting it to the research authorities. Dr. XCR, Dr. AMJ, Dr. CLDC, Dr. GII, Dr. JPAM, Dr. FVVP, Dr JJL, and Dr. AMA were in charge of the recruitment and operational follow-up of the cases. Dr. PEBI performed the statistical analysis and drafting of the manuscript. An external service translated the text into English.
| 1. | World Health Organization. WHO Coronavirus (COVID-19) Dashboard. 2021 [Cited 2021 Jul 16]. Available from URL: https://covid19.who.int |
| 2. | Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497-506. |
| 3. | Food and Drug Administration. Coronavirus (COVID-19) | Drugs. [Cited 2021 Jul 16]. Available from URL: https://www.fda.gov/drugs/emergency-preparedness-drugs/coronavirus-covid-19-drugs |
| 4. | European Medicines Agency. Science Medicines Health. COVID-19 medicines. [Cited 2021 Jul 16]. Available from URL: https://www.ema.europa.eu/en/human-regulatory/overview/public-health-threats/coronavirus-disease-covid-19/treatments-vaccines/treatments-covid-19/covid-19-treatments-authorised |
| 5. | Cecere TE, Todd SM, Leroith T. Regulatory T cells in arterivirus and coronavirus infections: Do they protect against disease or enhance it? Viruses. 2012. 4(59:833-46. |
| 6. | Fajgenbaum DC, June CH. Cytokine storm. N Engl J Med. 2020;383(23):2255-73. |
| 7. | Yeleswaram S, Smith P, Burn T, Covington M, Juvekar A, Li Y, et al. Inhibition of cytokine signaling by ruxolitinib and implications for COVID-19 treatment. Clin Immunol. 2020;218: 108517. |
| 8. | Guimarães PO, Quirk D, Furtado RH, Maia LN, Saraiva JF, Antunes MO, et al. Tofacitinib in patients hospitalized with Covid-19 pneumonia. N Engl J Med. 2021;385(5):406-15. |
| 9. | Kalil AC, Patterson TF, Mehta AK, Tomashek KM, Wolfe CR, Ghazaryan V, et al. Baricitinib plus remdesivir for hospitalized adults with Covid-19. N Engl J Med. 2021;384(9):795-807. |
| 10. | Marconi VC, Ramanan AV, de Bono S, Kartman CE, Krishnan V, Liao R, et al. Efficacy and safety of baricitinib for the treatment of hospitalised adults with COVID-19 (COV-BARRIER): A randomised, double-blind, parallel-group, placebo-controlled phase 3 trial. Lancet Respir Med. 202;9(12):1407-18. |
| 11. | Cantini F, Niccoli L, Matarrese D, Nicastri E, Stobbione P, Goletti D. Baricitinib therapy in COVID-19: A pilot study on safety and clinical impact. J Infect. 2020;81(2):318-56. |
| 12. | Cantini F, Niccoli L, Nannini C, Matarrese D, Di Natale ME, Lotti P, et al. Beneficial impact of baricitinib in COVID-19 moderate pneumonia; multicentre study. J Infect. 2020;81(4)647-79. |
| 13. | NOVARTIS. Novartis provides update on RUXCOVID study of ruxolitinib for hospitalized patients with COVID-19 | Novartis [Internet]. [Cited 2021 Sep 14]. Available from URL: https://www.novartis.com/news/media-releases/novartis-provides-update-ruxcovid-study-ruxolitinib-hospitalized-patients-covid-19 |
| 14. | Han MK, Antila M, Ficker JH, Gordeev I, Guerreros A, Lopez Bernus A, et al. Ruxolitinib in addition to standard of care for the treatment of patients admitted to hospital with COVID-19 (RUXCOVID): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Rheumatol. 2022;4(5):e351-e361. |
| 15. | Cao Y, Wei J, Zou L, Jiang T, Wang G, Chen L, et al. Ruxolitinib in treatment of severe coronavirus disease 2019 (COVID-19): A multicenter, single-blind, randomized controlled trial. J Allergy Clin Immunol. 2020;146(1):137-146.e3. |
| 16. | La Rosée F, Bremer HC, Gehrke I, Kehr A, Hochhaus A, Birndt S, et al. The Janus kinase 1/2 inhibitor ruxolitinib in COVID-19 with severe systemic hyperinflammation. Leukemia. 2020;34(7):1805-15. |
| 17. | Iastrebner M, Castro J, García Espina E, Lettieri C, Payaslian S, Cuesta MC, et al. Ruxolitinib in severe COVID-19: Results of a multicenter, prospective, single arm, open-label clinical study to investigate the efficacy and safety of ruxolitinib in patients with COVID-19 and severe acute respiratory syndrome. Rev Fac Cienc Med Univ Nac Cordoba. 2021;78(3):294-302. |
| 18. | Capochiani E, Frediani B, Iervasi G, Paolicchi A, Sani S, Roncucci P, et al. Ruxolitinib rapidly reduces acute respiratory distress syndrome in COVID-19 disease. Analysis of data collection from RESPIRE Protocol. Front Med (Lausanne). 2020;7:466. |
| 19. | Vannucchi AM, Sordi B, Morettini A, Nozzoli C, Poggesi L, Pieralli F, et al. Compassionate use of JAK1/2 inhibitor ruxolitinib for severe COVID-19: A prospective observational study. Leukemia. 2021;35(4):1121-33. |
| 20. | Ovilla-Martinez R, Cota-Rangel X, De La Peña-Celaya J, Alvarado-Zepeda MA, Jiménez Sastré A, Azuara Forcelledo H, et al. Ruxolitinib as a treatment strategy for SARS-CoV-2 pneumonia: Clinical experience in real-world setting. J Infect Dev Ctries. 2022;16(1):63-72. |
| 21. | Medscape. Jakafi. Ruxolitinib (Rx) [Internet]. [Cited 2020 Mar 26]. Available from URL: https://reference.medscape.com/drug/jakafi-ruxolitinib-999703 |
| 22. | Medscape. Xeljanz, Xeljanz XR. Tofacitinib (Rx) [Internet]. [Cited 2021 Sep 11]. Available from URL: https://reference.medscape.com/drug/xeljanz-xeljanz-xr-tofacitinib-999789 |
| 23. | Medscape. Olumiant. Baricitinib (Rx) [Internet]. [Cited 2021 Sep 11]. Available from URL: https://reference.medscape.com/drug/olumiant-baricitinib-1000107 |
| 24. | RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693-704. |
| 25. | Petrilli CM, Jones SA, Yang J, Rajagopalan H, O’Donnell L, Chernyak Y, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: Prospective cohort study. BMJ. 2020;369:m1966. |
| 26. | Bennett TD, Moffitt RA, Hajagos JG, Amor B, Anand A, Bissell MM, et al. Clinical characterization and prediction of clinical severity of SARS-CoV-2 infection among US adults using data from the US National COVID Cohort Collaborative. JAMA Netw Open. 2021;4(7):e2116901. |
| 27. | Dana R, Bannay A, Bourst P, Ziegler C, Losser MR, Gibot S, et al. Obesity and mortality in critically ill COVID-19 patients with respiratory failure. Int J Obes (Lond). 2021;45(9):2028-37. |
| 28. | Titanji BK, Farley MM, Mehta A, Connor-Schuler R, Moanna A, Cribbs SK, et al. Use of baricitinib in patients with moderate to severe coronavirus disease 2019. Clin Infect Dis. 2021;72(7):1247-50. |
| 29. | Ovilla-Martínez R, Cota-Rangel X, De La Peña-Celaya JA, Molina-Jaimes A, Alvarado-Zepeda MA, Rojas-Vértiz Contreras KE, et al. Ruxolitinib as treatment against COVID-19 in Mexican population. Rev Hematol Mex. 2020;21(4):195-204. |
| 30. | Dougan M, Nirula A, Azizad M, Mocherla B, Gottlieb RL, Chen P, et al. Bamlanivimab plus etesevimab in mild or moderate Covid-19. N Engl J Med. 2021;385(15):1382-92. |
| 31. | Razonable RR, Pawlowski C, O’Horo JC, Arndt LL, Arndt R, Bierle DM, et al. Casirivimab–imdevimab treatment is associated with reduced rates of hospitalization among high-risk patients with mild to moderate coronavirus disease-19. EClinicalMedicine. 2021;40:101102. |
| 32. | Food and Drug Administration. Emergency use authorization. [Retrieved on October 10th, 2023]. Available from URL: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#coviddrugs |
| 33. | National Institutes of Health. COVID-19 treatment and guidelines. Molnupiravir (Last updated: April 20, 2023). [Retrieved on October 10th, 2023. Available from URL: https://www.covid19treatmentguidelines.nih.gov/therapies/antivirals-including-antibody-products/molnupiravir/ |
| 34. | National Institutes of Health. Ritonavir-boosted Nirmatrelvir (paxlovid) (Last updated: October 10, 2023). [Retrieved on October 10th, 2023. Available from URL: https://www.covid19treatmentguidelines.nih.gov/therapies/antivirals-including-antibody-products/ritonavir-boosted-nirmatrelvir--paxlovid-/ |
All Rights Reserved® 2019
Latin American Journal of Clinical Sciences and Medical Technology,Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.
All Rights Reserved® 2019
Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.