| ** | Latin American Journal of Clinical Sciences and Medical Technology is an open access magazine. To read all published articles and materials you just need to register Registration is free of charge. Register now If you already have registered please Log In | ** |
aSección de Estudios de Posgrado e Investigación, Escuela Superior de Medicina, Instituto Politécnico Nacional; bDirección de Investigación, Instituto Nacional de Geriatría; cUnidad de Investigación en Farmacología, Instituto Nacional de Enfermedades Respiratorias, Ismael Cosio Villegas; dSección de Estudios de Posgrado e Investigación, Escuela Superior de Medicina, Instituto Politécnico Nacional, Unidad de Investigación en Farmacología, Instituto Nacional de Enfermedades Respiratorias, Ismael Cosio Villegas.
Corresponding Author: , . Telephone number: ; e-mail: juangreyesgarcia@gmail.com
Lat Am J Clin Sci Med Technol. 2025 Nov;7:90-99.
Received: September 11th, 2025.
Accepted: October 24th, 2025.
Published: November 4th, 2025.
Views: 2082
Downloads: 6
Background. Fenproporex/diazepam combination is prescribed as an adjuvant for the short-term treatment of obesity in Mexico and other Latin American countries, but the literature provides limited information on its effectiveness and safety. Objective. The current study performed a retrospective, multicenter, open-label, uncontrolled protocol to evaluate the effectiveness and safety of a fixed-dose combination of fenproporex/diazepam in adult overweight and obese patients from Mexico. Materials and Methods. One hundred eleven fenproporex/diazepam-treated overweight or obese patients were included in the current study. The primary outcome was the absolute body weight loss. The secondary outcomes included other anthropometric variables and cardiometabolic parameters, as well as the description of adverse events. Results. Fenproporex/diazepam decreased the absolute body weight by 7.1 ± 0.4 and 9.4 ± 0.6 kg at three and six months, respectively. Additionally, 82.0% and 55.0% of subjects achieved a weight loss of ≥5% or >10% at six months, respectively. Fenproporex/diazepam also decreased significantly body mass index,hip circumference, waist circumference, body fat, systolic blood pressure, and diastolic blood pressure, at three and six months. Mouth dryness, stress, anxiety, polyphagia, constipation, and insomnia were the most frequently reported adverse events. Conclusions. Data seem to indicate that the fenproporex/diazepam combination is effective and safe for up to six months, to treat weight loss in overweight and obese Mexican patients.
Antecedentes. La combinación de fenproporex/diazepam se prescribe como adyuvante para el tratamiento a corto plazo de la obesidad en México y otros países de Latinoamérica; sin embargo, la información sobre su efectividad y seguridad es limitada en la literatura. Objetivo. El presente estudio llevó a cabo un protocolo retrospectivo, multicéntrico, abierto y no controlado para evaluar la efectividad y seguridad de una combinación de dosis fija de fenproporex/diazepam en pacientes adultos mexicanos con sobrepeso y obesidad. Material y métodos. En el presente estudio se incluyeron 111 sujetos con sobrepeso u obesidad que fueron tratados con fenproporex/diazepam durante seis meses. La variable primaria de desenlace fue la pérdida absoluta de peso corporal, mientras que las variables secundarias incluyeron otras de tipo antropométrico y parámetros cardiometabólicos; así como la descripción de eventos adversos. Resultados. La administración oral de fenproporex/diazepam disminuyó el peso corporal absoluto en -7.1 ± 0.4 kg y -9.4 ± 0.6 kg, a los tres y seis meses, respectivamente. Además, 82.0% y 55.0% de los sujetos alcanzaron ≥5% o ≥10% de pérdida de peso a los seis meses, respectivamente. Fenproporex/diazepam también disminuyó significativamente el índice de masa corporal, la circunferencia de cadera, la circunferencia de cintura, el contenido de grasa corporal, la presión arterial sistólica y la presión arterial diastólica, a los tres y seis meses. Los eventos adversos reportados con mayor frecuencia fueron boca seca, estrés, ansiedad, polifagia, estreñimiento e insomnio. Conclusiones. Los datos sugieren que la combinación de fenproporex/diazepam es efectiva y segura para tratar la pérdida de peso en pacientes mexicanos con sobrepeso y obesidad por hasta seis meses.
Obesity is defined as an abnormal or excessive accumulation of fat. It is a significant risk factor for non-communicable diseases such as diabetes, cardiovascular disease, musculoskeletal disorders, and cancer, resulting in a dramatic decrease in quality of life.1,2 Furthermore, obesity is associated with a higher risk of all-cause mortality.3 Conversely, weight loss in obese individuals has beneficial effects on their health, including a decrease in the incidence of diabetes mellitus, sleep apnea, hypertension, dyslipidemia, asthma, and chronic kidney disease, among others.4,5
Obesity is a public health concern in Mexico, which has been increasing over the past 30 years. From 2000 to 2018, adult obesity increased by 42.2%. The 2018 National Health Survey reported that 36.1% of adults were obese, with a significantly higher prevalence in women (40.2% vs. 30.5%). Currently, the prevalence of adult obesity in the poorest regions of Mexico is similar to that in high-income areas. The leading causes of mortality in the country are associated with obesity, including cardiovascular disease (20.1%), type 2 diabetes (15.2%), malignant tumors (10.8%), and liver disease (7.6%).6
According to international and Mexican guidelines, pharmacotherapy should be considered as an adjuvant to diet and exercise in obese adults with a body mass index (BMI) of ≥30 kg/m2 or ≥27 kg/m2 and at least one comorbidity associated with excess adiposity.7
To date, in Mexico, there is a trend toward the use of drugs for long-term weight loss with the argument that obesity is a chronic disease; nevertheless, they still present adverse events that cannot be ignored.8,9 Their costs are usually much higher than those of short-term weight loss drugs, making pharmacological treatment of obesity affordable only for a small sector of the population.
Moreover, regardless of the treatment, the subjects will tend to develop tolerance to the drug, and they will regain the lost weight once it is discontinued if the subject fails to make lifestyle changes. In this regard, short-term weight loss drugs still have their place in therapy, but their safety and efficacy have been questioned due to a lack of clinical studies.
Fenproporex/diazepam is a fixed-dose anorectic combination prescribed in Mexico and other Latin American countries to treat obesity. It is suggested that fenproporex exerts its sympathomimetic effect in the brain, thereby inhibiting appetite.10-12
In controlled studies, fenproporex has shown to be significantly more effective than diet and exercise with weight reductions of 4.7 kg, 3.8 kg and 1.6 kg at 12, 6 and 2 months, respectively.13-16
Regarding its safety, the main concerns are its potential for abuse and the development of psychosis. Controlled clinical studies report insomnia, irritability, and anxiety as the main adverse events13-15, but its effectiveness or safety has been poorly studied in real-world clinical trials. Thus, the current phase IV clinical study was carried out to evaluate the effectiveness and safety of fenproporex/diazepam in overweight and obese Mexican subjects.
Patients and Methods
Study Design
To evaluate the effectiveness and safety of the administration of a fixed-dose combination of fenproporex/diazepam in overweight and obese Mexicans.
We debug a database with obese subjects pharmacologically trtheated with several weight loss drugs between July 2011 and April 2015. From the database, 111 patients treated with a daily extended-release capsule containing 20 mg fenproporex and 6 mg diazepam for six months and with complete anthropometric data were selected to participate in the current retrospective, uncontrolled, multicenter, phase IV open-label clinical trial.
The subjects included in the study were between 20 and 64 years old.
The protocol was approved by the Ethics Committee of INER/CEI/0113/15 (Code: C14-15).
From the initial visit, demographic and clinic data such as age (years), sex (woman or man), weight (kg), BMI (kg/m2), waist circumference (cm), hip circumference (cm), waist-to-hip ratio (WHR), body fat (kg, %), lean mass (kg), glucose (mg/dL), systolic blood pressure (SBP, mmHg), diastolic blood pressure (DBP, mmHg), heart rate (bpm), respiratory frequency (rpm), comorbidities, and habits (alcohol consumption, tobacco consumption, and sedentary lifestyle) were captured for the per-protocol analysis.
From the monthly follow-up visits, all adverse events reported by the physician were extracted and stratified by System Organ Class according to the Medical Dictionary for Regulatory Activities (MedDRA). In addition, the values of the clinical variables described in the initial visit were obtained monthly.
At the initial visit, clinical examination and medical history were used to establish the subjects' baseline health status.
Patients were advised to adhere to a 1500 kcal diet and engage in 20 minutes of physical activity each day. They were instructed to take one extended-release capsule containing 20 mg fenproporex and 6 mg diazepam daily 30 minutes before breakfast and return the following month for their follow-up visit.
In all visits, the subjects were weighed and measured in a bipedal position, in light clothes and without shoes by a calibrated scale and a stadiometer.
Waist circumference was measured at the level of the navel, whereas hip circumference was quantified at the fullest part of the buttocks using a flexible metric tape.
Heart rate, SBP and DBP values were got by a digital sphygmomanometer. Body fat and lean mass were measured by bioimpedance scale, while glucose was obtained using a glucometer.
In addition, in each follow-up visit, the physician recorded the adverse events reported by the subject through questioning.
Data Analysis
In the current study, a per-protocol analysis was performed.
The main endpoint was the absolute weight loss. Secondary endpoints comprised the number of subjects with at least 5% or 10% of weight loss, the reduction in the waist circumference, hip circumference, WHR, BMI and body fat, as well as, the change in lean mass, heart rate, SBP, DBP, glucose, respiratory rate and the frequency of adverse events. All variables were measured monthly during the pharmacological treatment.
Age, body weight, BMI, waist circumference, hip circumference, WHR, body fat, lean mass, glucose, SBP, DBP, heart rate and respiratory frequency are presented as mean ± standard deviation; whereas sex, obesity grades, comorbidities, alcohol consumption, tobacco consumption, sedentary life, 5% weight loss, 10% weight loss, and adverse events are presented as number of subjects and percentages.
Statistical differences in the anthropometric and clinic variables respect to their basal were obtained by one-way analysis of variance followed by Dunnett´s test. The statistical difference was significant when p <0.05. The creation of the figures and the statistical analysis was performed using GraphPad Prism 9.0.
Adverse events were classified according to System Organ Class proposed by MedDRA as cardiac cardiac, ear and labyrinth, eye, endocrine, gastrointestinal, musculoskeletal, connective tissue, nervous system, psychiatric, respiratory, thoracic, mediastinal, skin, and subcutaneous tissue disorders.
Demographic Data
Most of the patients were women (76.8%) between 30 and 39 years (31.5%), and obesity grade I (50.5%).
About a third of the subjects declared to be alcohol consumers (28.8%), a fifth tobacco consumers (22.5%), and half reported a sedentary lifestyle (52.3%).
Varicose vein (49.6%) was the most common comorbidity, followed by insomnia (29.7%), dyslipidemia (20.7%), and urinary incontinence (14.4%). Complete basal demographic and clinic data are shown in Table 1.
| Table 1. Baseline demographic and clinical variables of the subjects included in the 6 - month per - protocol analysis | |||||
|---|---|---|---|---|---|
| Variable | Number of subjects or mean | % or SD | Variable | Number of subjects or mean | % or SD |
| Age (years)a | 43.1 | 10.5 | Comorbiditiesb | ||
Sexb | Diabetes | 4 | 3.6 | ||
| Women | 108 | 97.3 | Dyslipidemia | 23 | 20.7 |
| Men | 3 | 2.7 | Hypertension | 5 | 4.5 |
Obesityb | Infertility | 4 | 3.6 | ||
| Overweight | 27 | 24.3 | Insomnia | 33 | 29.7 |
| Grade I | 56 | 50.5 | Gastroesophageal reflux disease | 18 | 16.2 |
| Grade II | 22 | 19.8 | Thyroid disease | 1 | 0.9 |
| Grade III | 6 | 5.4 | Urinary incontinence | 16 | 14.4 |
| Alcohol consumerb | 32 | 28.8 | Varicose veins | 55 | 49.6 |
| Tobacco consumerb | 25 | 22.5 | Body fat (%)a | 47.3 | 4.4 |
| Sedentary lifeb | 58 | 52.3 | Lean mass (kg)a | 23.4 | 8.7 |
| Weight (kg)a | 80.3 | 11.6 | Glucose (mg/dL)a | 96.6 | 17.3 |
| Body mass index (kg/m2)a | 32.8 | 4.0 | Systolic blood pressure (mmHg)a | 119.5 | 12.9 |
| Waist circumference (cm)a | 101.7 | 11.4 | Diastolic blood pressure (mmHg)a | 75.6 | 8.5 |
| Hip circumference (cm)a | 113 | 9.1 | Heart rate (lpm)a | 77.4 | 9.0 |
| Waist to hip ratioa | 0.9 | 0.1 | Respiratory rate (rpm)a | 17.4 | 2.5 |
| Body fat (kg)a | 10.3 | 3.3 | |||
| Data are presented as mean (standard deviation, SD)a or as the absolute number of patients (percentage)b | |||||
Fenproporex/diazepam Efficacy
Basal body weight was 80.3 ± 11.6 kg. The fixed dose of fenproporex/diazepam significantly reduces the basal weight to 73.2 ± 9.8 kg and 70.9 ± 9.5 kg, at three and six months, respectively, which corresponds to a percentage weight reduction of -8.6 ± 4.3% at three months and -11.3 ± 6.8% at six months. In addition, the average BMI decreased significantly from 32.8 ± 4.0 kg/m2 to 29.9 ± 3.3 kg/m2 and 29.0 ± 3.3 kg/m2 at three and six months, respectively (see Graphic 1).
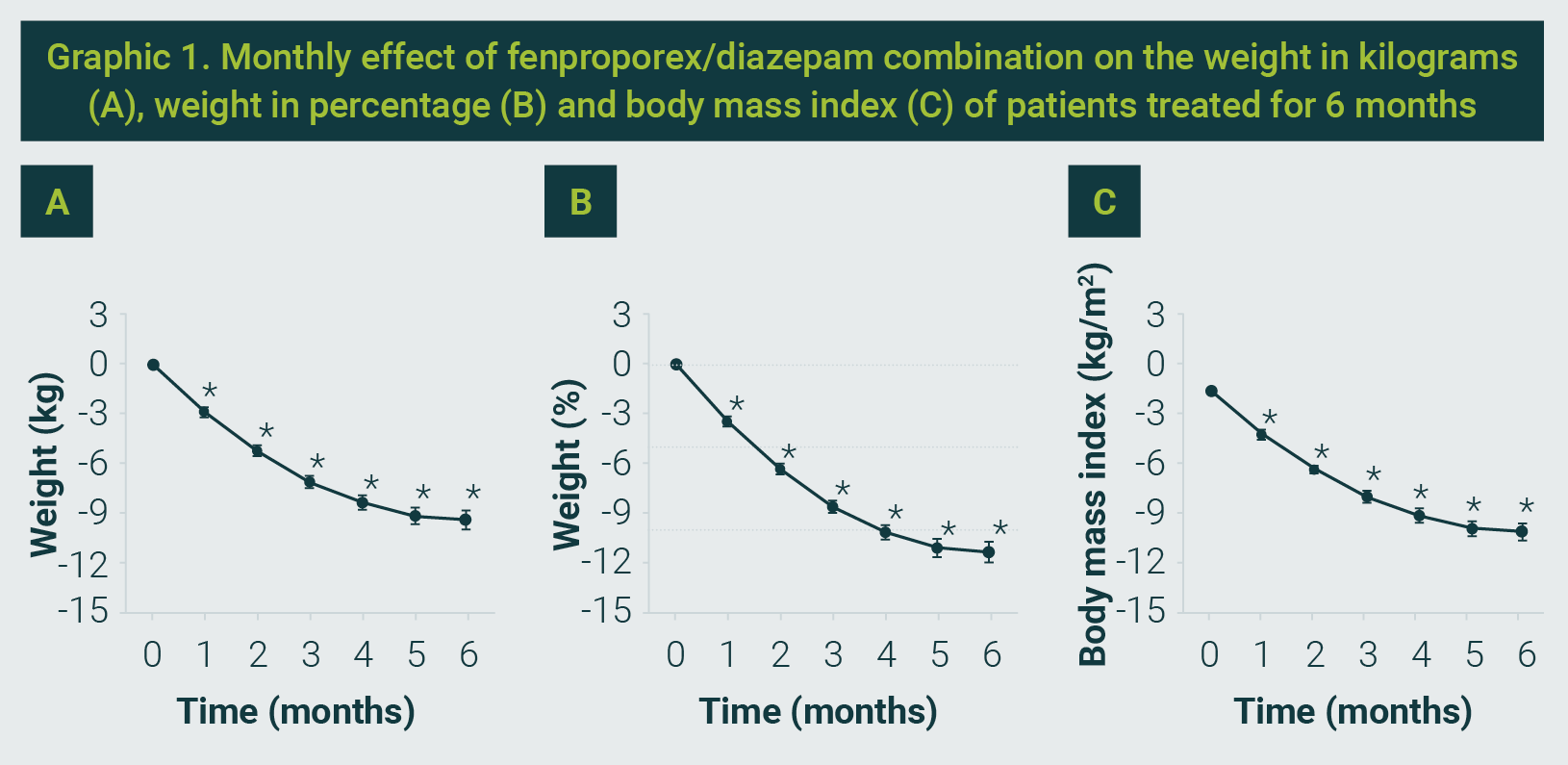
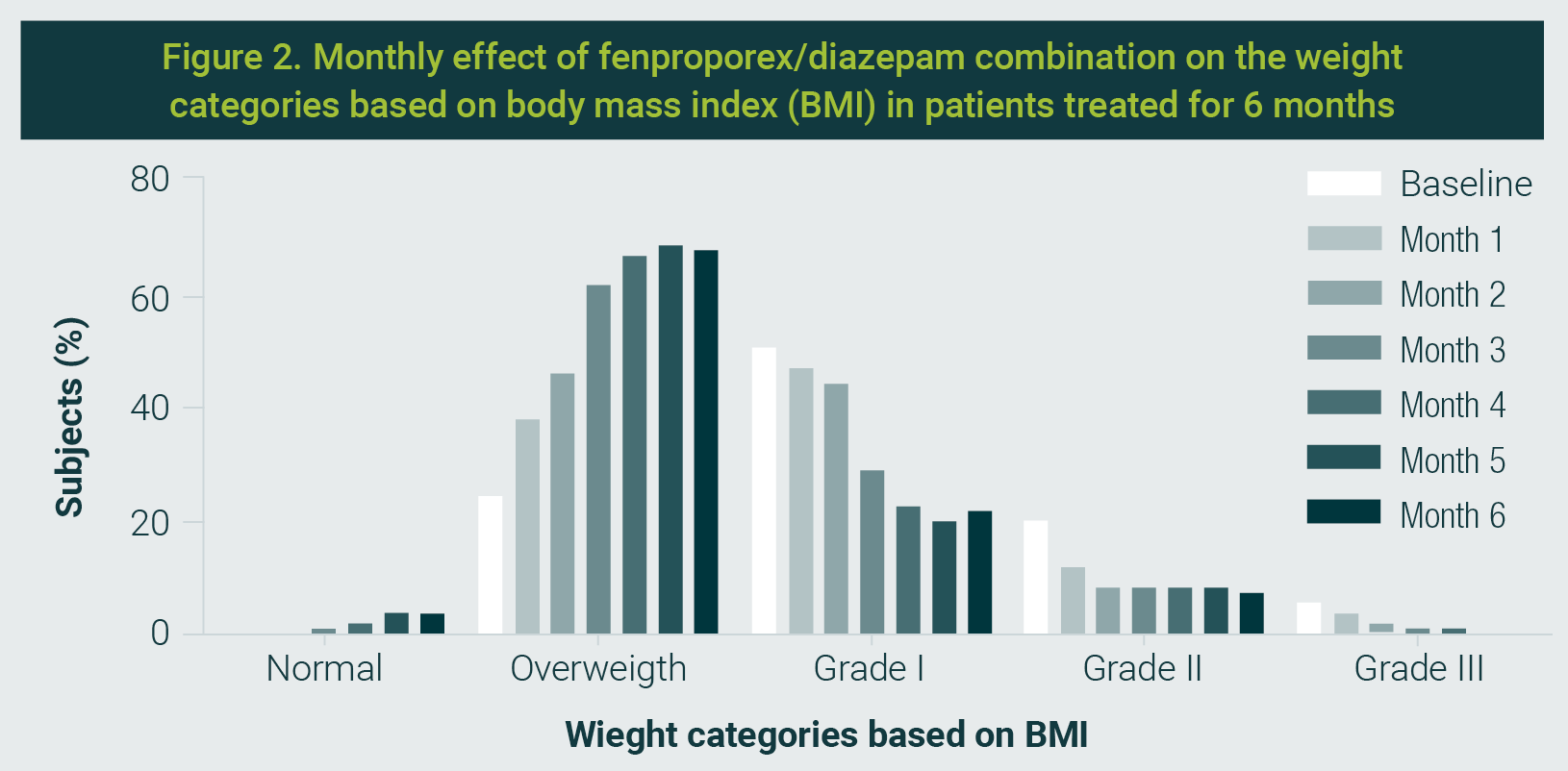
Within weight categories based on BMI, the percentage of subjects taking daily the fenproporex/diazepam combination increased in a time-dependent manner in normal weight (from 0% to 3.6%) and overweight (from 24.3% to 67.6%) categories, but it decreases in obese subjects of grade I (from 50.5% to 21.6%), grade II (from 19.8% to 7.2%) and grade III (from 5.4% to 0%) categories, see Graphic 2.
After three months, 83.8% and 34.2% of subjects achieved at least 5% and 10% body weight reduction, respectively. Similarly, 82.0% and 55.0% of subjects achieved at least 5% and 10% body weight loss, respectively, at the end of pharmacological treatment.
Likewise, the combination of fenproporex and diazepam significantly improved waist circumference, hip circumference, and body fat in the subjects included in the study at both time points analyzed.
Glucose levels tended to decrease with fenproporex/diazepam treatment, but the change was not statistically significant (Table 2).
| Table 2. Values obtained from overweight and obese patients who received a daily capsule of fenproporex / diazepam (20 mg / 6 mg) for 6 months | ||||||
|---|---|---|---|---|---|---|
| Characteristic (N = 111) | Month 1 | Month 2 | Month 3 | Month 4 | Month 5 | Month 6 |
| Body weight (≥ 5%) | 34 (30.6) | 76 (68.5) | 93 (83.8) | 95 (85.6) | 96 (86.5) | 91 (82.0) |
| Body weight (≥ 10%) | 2 (1.8) | 11 (9.9) | 38 (34.2) | 59 (53.2) | 65 (58.6) | 61 (55.0) |
| WC (cm) | -3.3±0.3* | -5.8±0.5* | -8.5±0.6* | -9.5±0.7* | -10.5±0.8* | -10.3±0.8* |
| HC (cm) | -2.8±0.4* | -4.8±0.4* | -7.1 ±0.4* | -8.2±0.5* | -8.8±0.6* | -9.0±0.7* |
| WHR | -0.007±0.003 | -0.014±0.004 | -0.019±0.004* | -0.020±0.004* | -0.024±0.004* | -0.021±0.005 |
| Body fat (%) | -1.2±0.3 | -2.4±0.3* | -3.4±0.3* | -4.5±0.5* | -5.0±0.6* | -4.9±0.4* |
| Body fat (kg) | -0.5±0.1 | -0.9±0.2* | -1.2±0.2* | -1.6±0.3* | -1.9±0.3* | -1.9±0.3* |
| Lean mass (kg) | -1.0±0.8 | -0.5±0.9 | -0.2±0.9 | 0.0±0.9 | 0.4±0.9 | 0.7±0.9 |
| Glucose (mg/dL) | -2.4±1.8 | -3.2±1.9 | -4.5±2.1 | -2.3±1.7 | -4.0±1.8 | -3.8±2.1 |
| SBP (mmHg) | -4.0±1.1 | -6.9±1.1* | -5.8±1.6* | -7.2±1.3* | -6.1±1.4* | -6.4±1.2* |
| DBP (mmHg) | -3.3±0.9* | -3.9±0.9* | -4.3±0.8* | -6.1±0.9* | -5.6±0.8* | -5.4±0.8* |
| Heart rate (bpm) | -1.4±1.1 | -2.1±1.1 | -4.8±1.2* | -2.9±0.9 | -2.1±1.0 | -3.2±0.9 |
| Respiratory rate (rpm) | -0.7±0.3 | -0.6±0.3 | 0.5±0.3 | 0.2±0.3 | 0.5±0.4 | 0.0±0.3 |
| Data represent the cumulative number of patients (%) or the mean change from baseline to 3 and 6 months±standard deviation. *Significantly different from respective baseline value (p <0.05), as determined by one-way analysis of variance, followed by the Dunnett`s test. DBP: diastolic blood pressure, HC: hip circumference, SBP: systolic blood pressure, WC: waist circumference, WHR: waist-to-hip ratio. | ||||||
Fenproporex/diazepam Safety
Regarding the cardiovascular system, the daily oral administration of fenproporex/diazepam was safe and well-tolerated for six months, as it significantly reduced SBP and DBP. Moreover, it induced a slight reduction in the heart rate, but without a statistical difference from baseline.
Likewise, fenproporex/diazepam did not significantly alter the respiratory frequency throughout the treatment, as shown in Table 2.
In the current study, 68.5% of subjects reported at least one adverse event. The most common disorders presented were the following: gastrointestinal (55.0%), psychiatric (51.4%), endocrine (31.5%), nervous system (27.9%), musculoskeletal, and connective tissue (20.7%).
Furthermore, headache (11.7%), depression (11.7%), skin striae (12.6%), insomnia (18.0%), constipation (23.4%), polyphagia (28.8%), anxiety (32.4%), dry mouth (42.3%) and stress (42.3%) were the adverse events that occurred in more than 10% of the subjects included in the study. No severe events were reported, but a patient reported suicidal ideation, which is considered a serious adverse event. Table 3 displays a summary of adverse events described during fenproporex/diazepam treatment.
| Table 3. Adverse events reported with fenproporex/diazepam (20 mg / 6mg) treatment. | |||||
|---|---|---|---|---|---|
| Adverse event | Frequency | % | Adverse event | Frequency | % |
| Patients with at least 1 adverse event | 76 | 68.5 | |||
Ear and labyrinth disorders | Eye disorders | ||||
| Tinnitus | 3 | 2.7 | Phosphenes | 2 | 1.8 |
| Total | 3 | 2.7 | Total | 2 | 1.8 |
Cardiac disorders | Skin and subcutaneous tissue disorders | ||||
| Chest pain | 1 | 0.9 | Acanthosis nigricans | 1 | 0.9 |
| Palpitations | 1 | 0.9 | Skin striae | 14 | 12.6 |
| Total | 2 | 1.8 | Total | 14 | 12.6 |
Endocrine disorders | Respiratory, thoracic and mediastinal disorders | ||||
| Diaphoresis | 1 | 0.9 | Cough | 5 | 4.5 |
| Nervousness | 2 | 1.8 | Dyspnea | 2 | 1.8 |
| Polydipsia | 4 | 3.6 | Expectoration | 4 | 3.6 |
| Polyphagia | 32 | 28.8 | |||
| Total | 35 | 31.5 | Total | 8 | 7.2 |
Gastrointestinal disorders | Nervous system disorders | ||||
| Abdominal pain | 7 | 6.3 | Dizziness | 7 | 6.3 |
| Constipation | 26 | 23.4 | Headache | 13 | 11.7 |
| Dry mouth | 47 | 42.3 | Insomnia | 20 | 18.0 |
| Nausea | 2 | 1.8 | Irritability | 1 | 0.9 |
| Pyrosis | 4 | 3.6 | Vertigo | 1 | 0.9 |
| Total | 61 | 55.0 | Total | 31 | 27.9 |
Musculoskeletal and connective tissue disorders | Psychiatric disorders | ||||
| Back pain | 10 | 9.0 | Anxiety | 36 | 32.4 |
| Neck pain | 1 | 0.9 | Depression | 13 | 11.7 |
| Pain in extremity | 10 | 9.0 | Personality disorder | 1 | 0.9 |
| Pain in hands | 1 | 0.9 | Stress | 47 | 42.3 |
| Weakness | 11 | 9.9 | Suicidal ideation | 1 | 0.9 |
| Total | 23 | 20.7 | Total | 57 | 51.4 |
| Data are presented as number de patients and percentages. | |||||
The present investigation examined the safety and efficacy of a fixed-dose combination of fenproporex (20 mg)/diazepam (6 mg), which is frequently used to treat obesity in Mexico and other Latin American countries.
Our findings indicate that the anthropometric variables decreased in a time-dependent manner with daily oral administration of an extended-release capsule containing fenproporex/diazepam over six months.
The results show that the combination of fenproporex and diazepam reduced body weight by 11.3 ± 6.8% (SD) after six months of treatment in our per-protocol analysis. Furthermore, 82% and 55% of subjects reached at least 5% and 10% of body weight loss, respectively; and the subjects diminished their BMI from obesity grade 1 (32.8 ± 4.0 kg/m2) to overweight (29.0 ± 3.3 kg/m2) on average; likewise, other anthropometric measures also decreased after a 6-month follow-up period.
In line with our results, Morín-Zaragoza et al. found that fenproporex/diazepam induced a -11.6% of weight loss after 180 days of treatment in their intention-to-treat analysis.13
In addition, 43% and 86% of subjects managed at least 5% and 10% of weight loss, respectively, and the subjects reduced their average baseline BMI from 41.3 kg/m2 (obese grade III) to 37.5 kg/m2 (obese grade II) at the end of treatment.
Remarkably, the authors showed that the fenproporex/diazepam combination was more effective than fenproporex alone in improving anthropometric variables such as body weight, BMI, hip circumference, and body fat content.
The superiority of the fenproporex/diazepam combination in efficacy could be explained by greater adherence to treatment, as diazepam reduces adverse events induced by fenproporex in the central nervous system, such as insomnia or somnolence.13
In other studies, a prospective, randomized, placebo-controlled clinical trial conducted in Brazil observed that weight loss with fenproporex (-7.8 ± 6.9 kg) was significantly greater than placebo (-3.1 ± 4.3 kg) by week 52 in the intent-to-treat population.14
In addition, a systematic review of controlled studies on the efficacy of fenproporex found that it induces additional body weight reductions of 4.7 kg (one year), 3.8 kg (six months), and 1.5 kg (two months), respectively, compared with the diet-and-exercise intervention.15
Thus, our results are in line with previous controlled clinical trials about the efficacy of fenproporex and extend this conclusion to a phase IV, open-label, uncontrolled study in Mexican subjects.
The current study provides evidence from obese Mexican patients that fenproporex/diazepam treatment, not only reduced anthropometric parameters in these subjects, but also showed a positive tendency to improve the glucose levels.
In addition, the fenproporex/diazepam combination significantly reduced body fat content without affecting lean mass. This finding is interesting because the literature suggests that sympathomimetics can cause lean mass loss by increasing thermogenesis and energy expenditure, leading to a loss of body weight —both fat and muscle.17
Concordantly, a previous clinical trial demonstrated that the fenproporex/diazepam combination improved the lipid profile after six months of treatment since it decreased triglycerides, total cholesterol, and low-density lipoproteins without modifying the high-density lipoproteins.13
The absence of lean mass loss in our study may be attributed to the inclusion of only patients who adhered to the pharmacological treatment. Thus, it is plausible that these patients also adhered to the physician's exercise instructions.
Despite the efficacy of fenproporex/diazepam as an effective drug, short-term weight loss drugs have been questioned in terms of safety.
Fenproporex is a sympathomimetic stimulant with a pharmacological profile similar to that of the amphetamines, which induces its anorexigenic effect by indirectly releasing norepinephrine.18
Since amphetamines increase blood pressure and heart rate,19 it has been suggested that fenproporex also has the potential to present cardiovascular risk, especially with high doses and prolonged use.
On the contrary, the results obtained in the current study indicated that the oral administration of fenproporex/diazepam for six months significantly decreased the blood pressure and showed a tendency to diminish the heart rate. Furthermore, only 1.8% of subjects presented any adverse event related to cardiac disorders.
These results are consistent with previous studies13,14 and confirm that fenproporex is well-tolerated at the cardiovascular system. Notwithstanding, fenproporex should be prescribed with caution in patients with controlled hypertension and be contraindicated in patients with cardiovascular diseases until studies in special groups are available.
Tachypnea has also been linked to the toxic effects of sympathomimetics.20 In our study, fenproporex/diazepam did not produce clinically significant changes in respiratory rate throughout treatment.
In the same way, some anorectics have been withdrawn from the market because they caused pulmonary arterial hypertension and valvular heart disease.21
No reports of pulmonary arterial hypertension or valvopathies were found in our database, nor is there evidence in the scientific literature that fenproporex produces these potentially fatal adverse events.
Today, it is known that fenfluramine and dexfenfluramine increase the risk of developing pulmonary arterial hypertension or valvular heart disease through the activation of the serotonergic system (5-HT2B receptors) more than the noradrenergic system.22
The results indicate that 68.5% of subjects reported at least one adverse event, all of which were mild or moderate, suggesting that the treatment was well tolerated in our population.
The organ system classes with the highest incidence of adverse events were gastrointestinal, psychiatric, endocrine, and nervous system disorders, with approximately one-third of treated subjects reporting at least one adverse event related to each of these systems.
Moreover, the adverse events that occurred in more than 15% of the study population were dry mouth, stress, anxiety, polyphagia, constipation, and insomnia. The adverse events reported in the study were expected and are consistent with the literature.13-15
There were no reports of drug abuse or addiction with the use of fenproporex/diazepam for six months in our database. Nonetheless, some published case reports clearly show drug misuse.
From the four case reports, the drug was obtained from an acquaintance in three cases, prescribed for a longer period than authorized by a practitioner in one case, taken in higher doses than recommended in three cases, and consumed for more than two years in three cases.
In all cases, subjects were able to successfully discontinue diet pill use, although in one case, the depression remained as a sequel.23-25
It is fair to say that the misuse of any medication brings an increased health risk, which should not always be considered as a definitive criterion for withdrawing a drug from the market. In addition, these drugs are controlled; therefore, their proper use requires a medical prescription and medical supervision.
Moreover, a patient reported suicidal ideation as a serious adverse event in our database. Still, its causality with pharmacological treatment is only probable or possible since this adverse event also occurs in patients with eating disorders without pharmacological treatment.
In general, weight loss drugs are accompanied by a warning regarding suicidal ideation, but the larger clinical trials have not demonstrated an increase in the suicide rate.26
In addition, the weight loss drugs are contraindicated in subjects with psychiatric disorders since these drugs exert their effect by modifying appetite centers in the central nervous system and may induce additional central nervous system effects. In this regard, obese people should be closely monitored for mental health concerns, regardless of the weight-loss drug used, to ensure that they receive the necessary support and care.
Although the present study provides clear evidence of the effectiveness of the fenproporex/diazepam combination when participants adhere to treatment, the efficacy of the drug treatment could be overestimated in real-world settings or in controlled clinical trials13-16, as subjects with incomplete anthropometric data or fewer than six visits were not included in the analysis.
Other limitations are the lack of a placebo group and the small sample size. Thus, controlled clinical studies with larger sample sizes are required to establish a better risk-benefit profile of fenproporex/diazepam as a long-term weight loss drug.
In conclusion, data show that the fixed-dose combination of 20 mg fenproporex/ 6 mg diazepam is effective in improving the body weight and other anthropometric and cardiometabolic parameters in obese Mexican subjects. Moreover, the results suggest that the fenproporex/diazepam combination is well-tolerated for up to six months.
Authors fully acknowlegde Productos Medix, S.A. de C.V., Mexico City, Mexico for the database given for this study.
None.
| 1. | Fruh SM. Obesity: Risk factors, complications, and strategies for sustainable long-term weight management. J Am Assoc Nurse Pract. 2017;29(S1):S3-S14. |
| 2. | Abdelaal M, le Roux CW, Docherty NG. Morbidity and mortality associated with obesity. Ann Transl Med. 2017;5(7):161. |
| 3. | Aune D, Sen A, Prasad M, Norat T, Janszky I, Tonstad S, et al. BMI and all-cause mortality: Systematic review and non-linear dose-response meta-analysis of 230 cohort studies with 3.74 million deaths among 30.3 million participants. BMJ. 2016;353:i2156. doi: |
| 4. | Cefalu WT, Bray GA, Home PD, Garvey WT, Klein S, Pi-Sunyer FX, et al. Advances in the science, treatment, and prevention of the disease of obesity: Reflections from a Diabetes Care Editors' Expert Forum. Diabetes Care. 2015;38(8):1567-82. |
| 5. | Haase CL, Lopes S, Olsen AH, Satylganova A, Schnecke V, McEwan P. Weight loss and risk reduction of obesity-related outcomes in 0.5 million people: Evidence from a UK primary care database. Int J Obes (Lond). 2021;45(6):1249-58. |
| 6. | Barquera S, Rivera JA. Obesity in Mexico: Rapid epidemiological transition and food industry interference in health policies. Lancet Diabetes Endocrinol. 2020;8(9):746-7. |
| 7. | Chávez-Manzanera EA, Vera-Zertuche JM, Kaufer-Horwitz M, Vázquez-Velázquez V, Flores-Lázaro JR, Mireles-Zavala L, et al. Mexican clinical practice guidelines for adult overweight and obesity management. Curr Obes Rep. 2024;13(4):643-66. |
| 8. | Idrees Z, Cancarevic I, Huang L. FDA-approved pharmacotherapy for weight loss over the last decade. Cureus. 2022;14(9):e29262. |
| 9. | Farzam K, Patel P. Tirzepatide. [Retrieved on October 27th, 2025]. 2024. Available from URL: https://www.ncbi.nlm.nih.gov/books/NBK585056/ |
| 10. | Cody JT, Valtier S. Detection of amphetamine following administration of fenproporex. J Anal Toxicol. 1996;20(6):425-31. |
| 11. | Kraemer T, Theis GA, Weber AA, Maurer HH. Studies on the metabolism and toxicological detection of the amphetamine-like anorectic fenproporex in human urine by gas chromatography-mass spectrometry and fluorescence polarization immunoassay. J Chromatogr B Biomed Sci Appl. 2000;738(1):107-18. |
| 12. | de Cássia Mariotti K, Pechansky F, do Carmo Vieira Duarte P, et al. Fenproporex and amphetamine pharmacokinetics in oral fluid after controlled oral administration of fenproporex. Ther Drug Monit. 2012;34(5):545-53. |
| 13. | Morín Zaragoza R, Ponce López ML, Lonngi Villanueva S, Alcaraz Ortiz R, Lonngi Villanueva G. Eficacia y seguridad de dos formulaciones de fenproporex de liberación lenta como tratamiento de obesidad. Rev Mex Cardiol. 2005;16(4):146-54. [Consultado el 27 de octubre de 2025]. Disponible en URL: https://www.medigraphic.com/pdfs/cardio/h-2005/h054b.pdf |
| 14. | Suplicy H, Boguszewski CL, dos Santos CM, do Desterro de Figueiredo M, Cunha DR, Radominski R. A comparative study of five centrally acting drugs on the pharmacological treatment of obesity. Int J Obes (Lond). 2014;38(8):1097-103. |
| 15. | Paumgartten FJ, Pereira SS, de Oliveira AC. Safety and efficacy of fenproporex for obesity treatment: a systematic review. Rev Saude Publica. 2016;50:25. |
| 16. | Garcia Ramirez AV, Ribas Filho D, Zotarelli Filho IJ. Meta-analysis and approach of the real impact of anorexigenic drugs in the obesity in humans: The last five years of the randomized studies. Curr Diabetes Rev. 2020;16(7):750-8. |
| 17. | Christoffersen BØ, Sanchez-Delgado G, John LM, Ryan DH, Raun K, Ravussin E. Beyond appetite regulation: Targeting energy expenditure, fat oxidation, and lean mass preservation for sustainable weight loss. Obesity (Silver Spring). 2022;30(4):841-857. |
| 18. | Teodorak BP, Ferreira GK, Scaini G, Wessler LB, Heylmann AS, Deroza P, et al. Acute administration of fenproporex increased acetylcholinesterase activity in brain of young rats. An Acad Bras Cienc. 2015;87(2 Suppl):1389-95. |
| 19. | Chan M, Chan JJ, Wright JM. Effect of amphetamines on blood pressure. Cochrane Database Syst Rev. 2025;3(3):CD007896. |
| 20. | Schaffer DH, Richards JR. Sympathomimetic toxicity. [Retrieved on October 28th, 2025]. 2025. Available from URL: https://www.ncbi.nlm.nih.gov/books/NBK430757/ |
| 21. | Vivero LE, Anderson PO, Clark RF. A close look at fenfluramine and dexfenfluramine. J Emerg Med. 1998;16(2):197-205. |
| 22. | Bender AM, Parr LC, Livingston WB, Lindsley CW, Merryman WD. 2B determined: The future of the serotonin receptor 2B in drug discovery. J Med Chem. 2023;66(16):11027-11039. |
| 23. | Pélissier-Alicot AL, Piercecchi-Marti MD, Bartoli C, Kuhlmann E, Coiffait PE, Sanvoisin A, et al. Abusive prescription of psychostimulants: A study of two cases. J Forensic Sci. 2006;51(2):407-10. |
| 24. | Cohen PA. Imported fenproporex-based diet pills from Brazil: A report of two cases. J Gen Intern Med. 2009;24(3):430-3. |
| 25. | Smith BR, Cohen PA. Dependence on the Brazilian diet pill: A case report. Am J Addict. 2010;19(3):291-2. |
| 26. | Hoofnagle JH. Weight loss agents. In: LiverTox: Clinical and research information on drug-induced liver injury. 2025. [Retrieved on October 28th, 2025] Available from URL: https://www.ncbi.nlm.nih.gov/books/NBK548929/ |
All Rights Reserved® 2019
Latin American Journal of Clinical Sciences and Medical Technology,Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.
All Rights Reserved® 2019
Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.