| ** | Latin American Journal of Clinical Sciences and Medical Technology is an open access magazine. To read all published articles and materials you just need to register Registration is free of charge. Register now If you already have registered please Log In | ** |
aDepartment of Pharmaceutical Sciences and Technology, Faculty of Chemical and Pharmaceutical Sciences, University of Chile & Laboratory of Chemical Carcinogenesis and Pharmacogenetics, Department of Basic-Clinical Oncology, Faculty of Medicine, University of Chile, Santiago, Chile; bFaculty of Medical Sciences, Bernardo O´Higgins University, Santiago, Chile.
Corresponding Author: , . Telephone number: ; e-mail: lquinone@uchile.cl
Lat Am J Clin Sci Med Technol. 2026 Mar;8:14-24.
Received: January 16th, 2026.
Accepted: January 30th, 2026.
Published: March 2nd, 2026.
Views: 830
Downloads: 6
Background. Respiratory symptoms, such as cough, are frequent causes of medical consultation and are commonly managed with both synthetic and phytotherapeutic products. While several herbal components have been individually documented to have pharmacological properties, mixed phytotherapeutic formulations for respiratory symptoms have been less systematically evaluated. Objectives. To summarise the scientific background supporting the individual components of a multi-herbal phytotherapeutic syrup and to assess physicians’ and adult users’ perceptions regarding its effectiveness and tolerability in real-world use. Materials and Methods. A narrative review of the pharmacological and clinical evidence related to a formulation of eucalyptus leaf extract, avocado leaves, anise seeds, and honey was conducted. In parallel, two anonymised, structured perception surveys adapted from validated instruments were administered to practising physicians and adult users in Chile. Data were analysed using descriptive statistics. Results. A total of 144 physicians and 394 users completed the surveys: 96,5% of physicians were aware of the product, and 83,6% had prescribed it, mainly for a productive cough. Overall effectiveness was rated as moderate to high by 87,1% of physicians, and tolerability was considered good or excellent by 81,4%, with infrequent mild adverse events. Users reported perceived benefits in cough reduction (93,9%), mucus clearance (91,5%), relief of throat discomfort (93,1%), and overall improvement in respiratory symptoms (88,1%), while 97,9% reported no adverse effects. Conclusions. The reviewed botanical components have been individually documented to have anti-inflammatory, expectorant, and demulcent properties, consistent with perceptions reported by physicians and users. These findings, based on descriptive survey data and narrative evidence, suggest that mixed phytotherapeutic formulations warrant further investigation. Controlled clinical studies are required to establish safety and effectiveness under rigorous experimental conditions.
Antecedentes. Los síntomas respiratorios, como la tos, son causa frecuente de consulta médica y a menudo se tratan con productos sintéticos y fitoterapéuticos. Si bien, se ha documentado que, por separado, varios ingredientes herbales tienen propiedades farmacológicas, las formulaciones fitoterapéuticas mixtas para tratar síntomas respiratorios han sido menos evaluadas de modo sistemático. Objetivos. Resumir los antecedentes científicos que apoyan el uso de los ingredientes individuales de un jarabe fitoterapéutico multiherbal; y evaluar las percepciones de los médicos y de los usuarios del jarabe con respecto a la efectividad y tolerabilidad del uso real de dicho jarabe. Material y métodos. Se llevó a cabo una revisión narrativa de la evidencia farmacológica y clínica de una formulación de extracto de hoja de eucalipto, hojas de aguacate, semillas de anís y miel. Al mismo tiempo, se aplicaron dos encuestas de percepción anónimas y estructuradas (adaptadas de instrumentos validados) a médicos tratantes y a adultos usuarios del jarabe en Chile. Se analizaron los datos mediante estadística descriptiva. Resultados. Un total de 144 médicos y 394 consumidores completaron las encuestas. Entre los médicos, 96,5% sabía de la existencia del producto y 83,6% lo había prescrito, principalmente para tos productiva. El porcentaje de médicos que tasó la efectividad global de moderada a alta fue de 87,1%; y 81,4% consideró que la tolerabilidad fue buena o excelente y que los eventos adversos fueron poco frecuentes y moderados. Los usuarios refirieron haber percibido disminución de la tos (93,9%), eliminación de mucosidad (91,5%), alivio de las molestias en la garganta (93,1%) y disminución global de los síntomas respiratorios (88,1%); 97,9% no refirió eventos adversos. Conclusiones. Se habían documentado, de modo individual, las propiedades antinflamatorias, expectorantes y emolientes de los ingredientes botánicos revisados, lo cual coincide con las percepciones que reportaron los médicos y los usuarios del jarabe. Estos hallazgos, basados en los datos de la encuesta descriptiva y en la evidencia narrativa, sugieren que las formulaciones fitoterapéuticas mixtas justifican la pertinencia de mayor investigación. Se requieren estudios clínicos controlados para establecer la seguridad y efectividad bajo condiciones experimentales rigurosas.
The human respiratory system processes approximately 12,000 litres of air per day, enabling essential gas exchange but also exposing the lungs to harmful agents such as microorganisms, irritant gases, particulate matter, allergens, medications, foreign bodies, and radiation. These can enter primarily via inhalation or aspiration, and less frequently through internal pathways.
Defence mechanisms include physical barriers, mucus production, cough, macrophages, and antimicrobial molecules.
Cough is a protective reflex that expels harmful substances from the airways. However, when persistent, it may lead to physical complications and exacerbate underlying diseases, impairing quality of life.1,2
Management ranges from traditional herbal preparations to synthetic medicines. Infusions containing honey with lemon, ginger, ivy, and eucalyptus have long been used for their expectorant and soothing properties, while steam inhalation with thyme can promote airway clearance.
Synthetic medicines such as
- dextromethorphan suppress dry cough;
- expectorants like guaifenesin facilitate mucus clearance in productive cough;
- antihistamines may be useful in allergic causes; and
- bronchodilators are essential for obstructive conditions such as asthma and chronic obstructive pulmonary disease (COPD).3-7
Physiology, Aetiology, and Classification of Cough
Cough is an essential reflex that protects the respiratory tract by expelling foreign material and secretions. It may be voluntary or involuntary, triggered by mechanical or chemical stimulation of receptors in the bronchial tree.8
Effectiveness depends on the coordination of the cough reflex, adequate expiratory force, and intact mucociliary function. While usually transient, it may cause complications such as headache, tussive syncope, arterial dissection, splenic rupture, insomnia, and anxiety.9
By duration, cough is classified as
- acute (<3 weeks),
- subacute (3-8 weeks), or
- chronic (>8 weeks).10
Common causes include viral respiratory infections, asthma, COPD, bronchiectasis, and pneumonia.
Subacute cough is often post-infectious, whereas chronic cough is associated with upper airway cough syndrome (UACS), non-asthmatic eosinophilic bronchitis (NAEB), gastro-oesophageal reflux disease (GORD), and cough-variant asthma.
The cough reflex protects the lower airways by preventing the entry of harmful particles (Figure 1). It is triggered when mucosal receptors detect irritants, initiating three phases: deep inspiration, compression with glottic closure, and rapid air expulsion.11-13
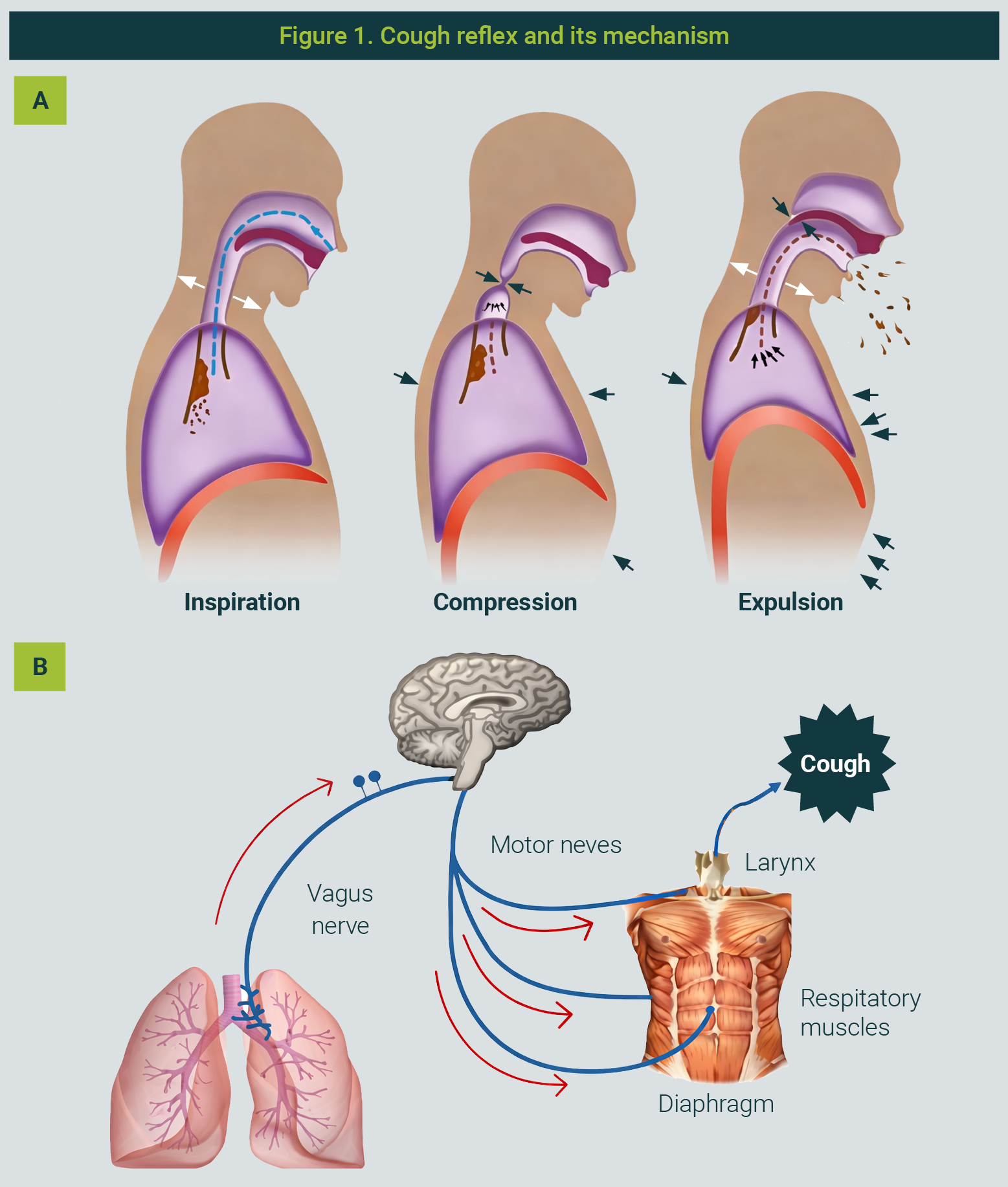
The figure is property of the authors
Receptors are located in the larynx, trachea, and bronchi, including rapidly adapting receptors (RAR), slowly adapting receptors (SAR), and C-fibre nociceptors.12 These respond to chemical stimuli via TRPV1 and TRPA1 channels, releasing neuropeptides that induce bronchoconstriction and secretion.
Sensory input is processed in the nucleus tractus solitarius and cortex, allowing partial voluntary control, while the efferent pathway activates respiratory muscles, the larynx, and pelvic sphincters during cough.13,14
Acute and subacute cough are often associated with upper respiratory tract infections such as colds, influenza, acute sinusitis, and bronchitis, as well as with asthma or COPD exacerbations, or with irritants such as tobacco smoke, and chemicals.
Viral infections can sensitise cough receptors, prolonging symptoms post-recovery.
Chronic cough is more commonly linked to UACS, GORD, asthma, NAEB, COPD, and use of certain medications such as angiotensin-converting enzyme (ACE) inhibitors, which cause persistent cough in up to 20% of patients.10,14,15
By character, cough may be dry or productive. A productive cough facilitates mucus clearance, while a dry cough is often more irritative. Expectorated sputum may occasionally contain blood (haemoptysis) or vary in appearance depending on the underlying condition.
Cough Treatment
Management depends on aetiology and may involve synthetic or natural medicines acting at central, peripheral or mixed levels16-29 (Table 1).
| Table 1. Allopathic and natural medicines for the management of human cough | |||
|---|---|---|---|
| Product | Mechanism of action | Indication | |
Allopathic | |||
Central action | Codeine Dextromethorphan Noscapine Carbetapentane Caramiphen Clofedanol Glaucine Butamirate Oxeladine | Inhibition of the cough centre stimulus, preventing the cough reflex | Conditions accompanied by little or no secretion in the airways |
Peripheral action | Levodropropizine Lidocaine Benzocaine Ipratropium | Inhibition of the stimulus on cough receptors | All types of coughs, except those originating from stimuli outside the respiratory system |
Mixed action | Cloperastine | Inhibition of stimulus at both the cough centre and receptors | All types of coughs |
| Diphenhydramine | |||
Others | N-acetylcysteine (NAC) | Breaks the bond of mucoproteins, reducing the viscosity of pulmonary secretions | Productive cough associated with benign bronchial conditions; used as an expectorant (coadjuvant) |
| Vitamins (C, E, B, etc.) | Antioxidant and immunostimulant effect | Relief of cough symptoms (coadjuvant) | |
Natural* | |||
| Product | Suggested mechanism of action | Suggested indication | |
| Extracts or infusionsof Hedera helix | Anti - inflammatory action related to inhibition of nuclear factor κB (NF-κB) and interleukin-6 (IL-6) release | Productive cough associated with benign bronchial conditions; expectorant | |
| Extracts or infusions of Eucalyptus globulus | Inhibit histamine release, improves lung compliance, and inhibits production of pro - inflammatory mediators | Bronchitis and pharyngitis | |
| Extracts or infusions of Echinacea purpurea | Stimulate the immune system, enhances phagocytosis, and may inhibit microbial enzymes | Common colds, flu, bronchitis, sinusitis | |
| Propolis | Anti-inflammatory, antioxidant, and antimicrobial effects | Prevention and treatment of respiratory tract diseases mainly caused by viruses | |
| Honey (alone or with lemon) | Coating of cough receptors, stimulation of saliva production, and reduction of inflammation in the airways | General relief of cough and sore throat | |
| Extracts or infusions of ginger (Zingiber officinale) | Mechanisms involving relaxation of airway smooth muscle, reduced inflammation, and modulation of intracellular calcium | Colds and general cough | |
| Marshmallow root extracts (Althaea officinalis) | Form a protective mucilaginous layer on irritated airways, reducing inflammation and inhibiting cough reflex via serotonergic receptors | Sore throat | |
| Extracts or infusions of thyme (Thymus vulgaris) | Components such as thymol and carvacrol act as antispasmodics (β2-receptors), expectorants, and enhance mucociliary activity | General relief of cough and sore throat | |
| Extracts or infusions of pineapple (Ananas comosus) | Anti-inflammatory and expectorant activity due to its enzyme bromelain | General cough and congestion | |
| Extracts or infusions of slippery elm (Ulmus rubra) | Anti-inflammatory and antioxidant effects; mucilage content coats and soothes the throat | General cough and throat irritation | |
| Extracts or infusions of avocado leaves (Persea americana) | Antiseptic and emollient properties | Relief of cough and symptoms of colds and flu | |
| *Natural, handcrafted, or pharmaceutical grade. Note. When “extract” is mentioned, it refers to the possibility that it may be either a crude extract or a standardised extract (for one or more components).16-29 The table is property of the authors. | |||
The treatment includes:
- Antitussives, which inhibit the cough reflex and are recommended for dry cough only.
- Expectorants, which stimulate mucus clearance in hypersecretory conditions, though their efficacy remains debated.30
- Mucolytics such as acetylcysteine and carbocisteine, which reduce mucus viscosity to facilitate expulsion.31
- Bronchodilators, which relax bronchial smooth muscle, especially in asthma and COPD.32
Non-pharmacological measures include positive expiratory pressure (PEP) therapy, mechanically assisted cough, and high-frequency chest oscillation, useful in chronic conditions such as cystic fibrosis and COPD.31
Home strategies include avoiding environmental pollutants, maintaining a healthy diet, using humidifiers or steam vaporizers, engaging in physical exercise, and nasal irrigation with saline or seawater.28
Paltomiel® is a syrup indicated as an adjuvant in productive cough, widely used in Chile in adults and children over 2½ years of age. It contains extracts of Eucalyptus globulus, Persea americana leaves, and Pimpinella anisum seeds (the latter absent in the paediatric formula), with honey as a demulcent to enhance respiratory benefits.
Although it may interact with anticoagulants, antidiabetics, and monoamine oxidase inhibitors, and is contraindicated in severe liver disease or hypersensitivity to its components33, its use is primarily based on traditional knowledge, as no clinical trials have evaluated its efficacy as a mixed formulation. However, its individual components have strong scientific support and are included in medical patents for antimicrobial, expectorant, anti-inflammatory, and antitussive properties.
In the Chilean market, most herbal cough syrups are based on Hedera helix (ivy), but Paltomiel® differs in its unique combination of active ingredients, offering a distinct therapeutic profile for productive cough.
This study aimed to describe its composition, review the scientific evidence available for its components, and assess the perceptions of physicians and users regarding its effectiveness and tolerability, considering its decades-long presence in the national market.
To gather data on the use of mixed formulations of eucalyptus leaf extract and/or avocado leaves and/or anise seeds and/or honey and similar products. We evaluated Paltomiel®, and two structured surveys were designed: one for the target public (users) and another for practising physicians in Chile.
Sample Size Calculation
Sample size was determined using the IdSurvey eucalyptus leaf extract, avocado leaves, anise seeds, and honey using a simple-size calculator (https://www.idsurvey.com/es/simple-size-calculator/), assuming a 5% margin of error, 95% confidence level, standard deviation of 50, a 5% attrition rate, and a total population of approximately 20 million potential users in Chile.
For the medical group, an estimated population of 1,200 physicians was considered, based on the 2024 report of the Chilean Health Superintendency regarding medical specialties.34 This calculation yielded statistically significant sample sizes of 144 physicians and 385 users.
Survey Administration
Participants were contacted to complete an anonymous, structured questionnaire evaluating their perception of the product’s safety and effectiveness.
Surveys were designed in-house but adapted from established, validated instruments, including the Treatment Satisfaction Questionnaire for Medication (TSQM) and the Medication Satisfaction Questionnaire (MSQ), which assess patient perception of the effectiveness and satisfaction with medical products.35,36
Survey Structure
The physicians’ survey was divided into five sections:
- Brief description of the survey’s scope.
- General physician data (speciality, years of experience, product awareness) - anonymous.
- Use and prescription of the product.
- Product evaluation (effectiveness, toxicity, tolerability).
- General opinion (recommendations and suggestions).
The users’ survey also comprised five sections:
- Brief description of the survey’s scope.
- General user data (age, sex, product awareness) - anonymous.
- Product evaluation (awareness, effectiveness, toxicity, tolerability).
- Impact of the product on well-being.
- Overall perception (other effects, satisfaction, comments).
Certain questions allowed for open-text responses, while others employed a 1-to-5 rating scale.
The research team developed draft versions of both questionnaires, based on items from the TSQM and the MSQ.
Face and content validity were assessed by two healthcare professionals with expertise in respiratory medicine and phytotherapeutic products, who independently reviewed the wording, relevance, and completeness of all items. Their comments were incorporated to refine the questionnaires before their online dissemination.
Statistical Analysis
Data from the physicians’ and users’ questionnaires were exported to a spreadsheet and analysed using descriptive statistics. All variables were categorical and are presented as absolute and relative frequencies (%). Missing data were treated as “no response” (NR) and reported as such in the results, tables, and figures. No inferential statistical tests or data imputation procedures were performed.
Ethical Considerations
This study used an anonymous, voluntary online survey and did not collect personally identifiable data. In accordance with national regulations for non-interventional survey-based research, formal ethics committee approval was not required.
All participants provided electronic informed consent before completing the questionnaire and were informed of the study's objectives, the voluntary nature of participation, and the confidentiality of their responses.
The surveys were conducted between December 29th, 2024, and March 6th, 2025, targeting physicians and adult users (>18 years) via various direct and digital channels. A total of 144 physicians and 394 users participated.
Physicians’ Perception
Professional profile
Most respondents specialised in general medicine (69,4%), followed by paediatrics (9,0%), family medicine (4,9%), geriatrics (2,8%), and pneumology (2,1%). A further 11,8% represented other specialties less directly associated with respiratory diseases. Regarding professional experience, 67% had more than 10 years of practice, 15% had 5-10 years, and 18% had fewer than 5 years.
Product Awareness and Prescription
Paltomiel® was known to 96,5% of physicians, and 83,6% had prescribed it—either occasionally (52,9%) or regularly (30,7%). Among non-prescribers, the main reasons cited were a preference for other treatments (63,6%), infrequent management of respiratory cases, or uncertainty regarding the efficacy of these treatments (Figure 2).
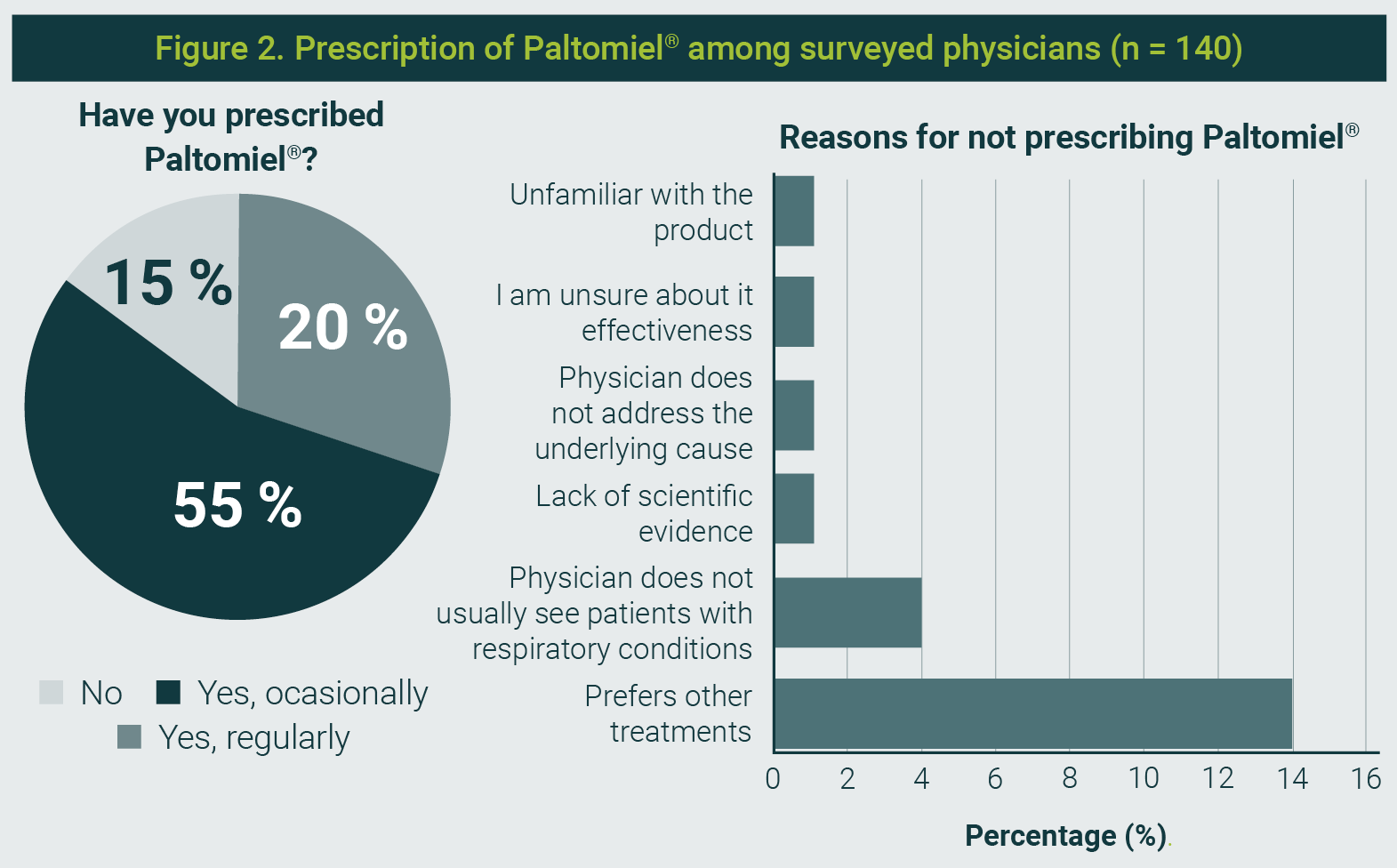
The figure is property of the authors
Perceived Effectiveness
- For a productive cough, 80% rated the syrup as very effective or extremely effective.
- For dry cough, 75% gave a positive rating, most frequently as moderately effective.
- For respiratory congestion, 67,1% considered it effective, with 29,3% describing it as moderately effective.
Overall effectiveness was rated between moderately and extremely effective by 87,1% of respondents, with the highest category (extremely effective) selected by 33,6% (Figure 3).
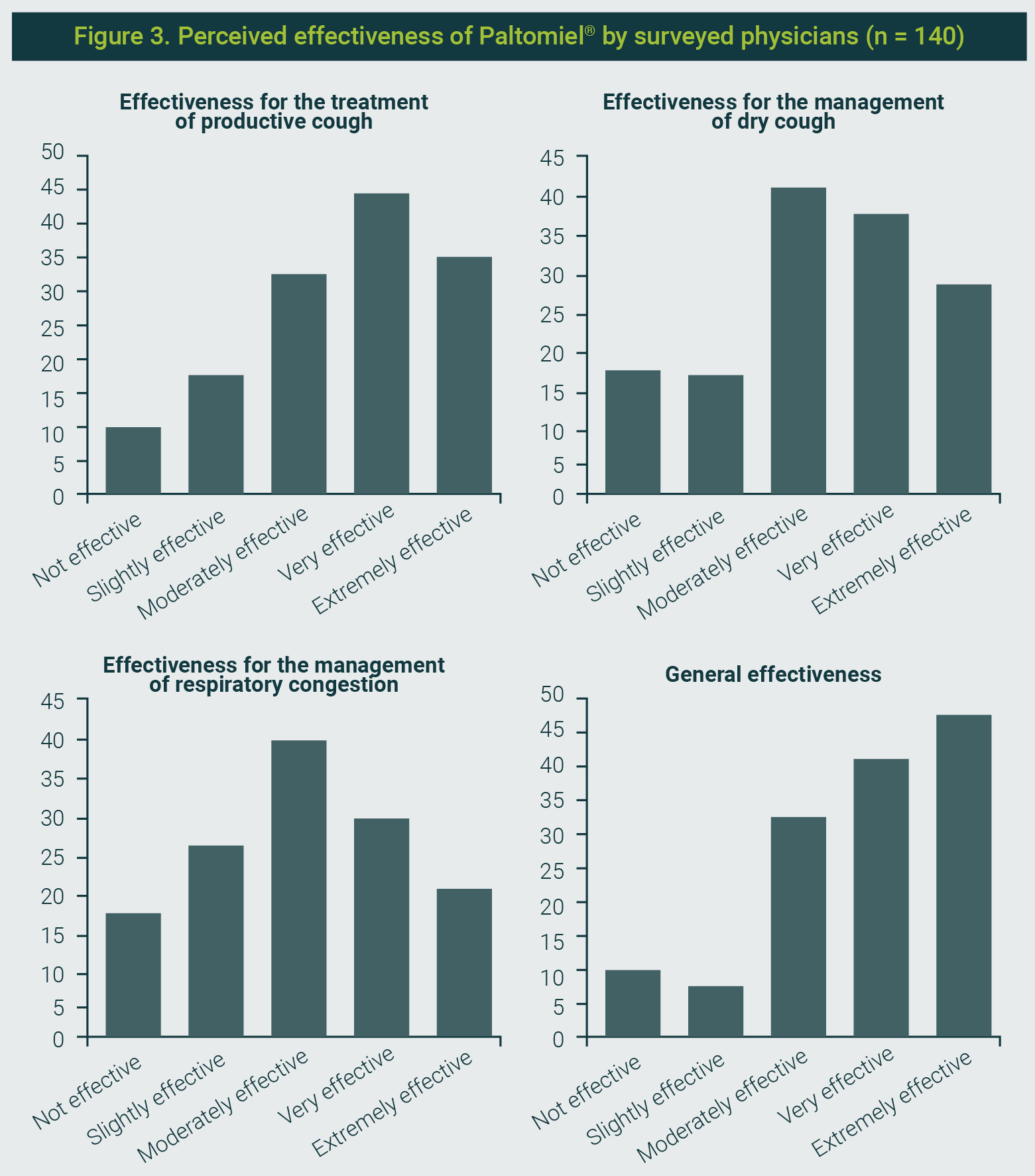
The figure is property of the authors
Tolerability and Safety
Organoleptic tolerability (taste, sweetness, consistency) was assessed as good or excellent by 81,4% of physicians, and no reports of poor tolerability were recorded. Most (96,4%) reported no relevant adverse effects, with only four cases of mild diarrhoea.
Intention to Continue Prescribing the Syrup
A total of 80,7% stated they were very likely or likely to continue prescribing the product, and 80% would recommend it to colleagues. Among those who did not recommend it (17,9%), the most common reasons were the absence of specific scientific evidence and limited demonstrated efficacy.
Users’ Perception
Demographics and Respiratory History
Among 394 respondents, 65,4% were female, with a mean age of 43,5 ± 14,2 years. The most frequently reported respiratory conditions were bronchitis (62,4%), asthma (13,7%), pneumonia (6.6%), and COPD (1,0%).
Product Awareness
A total of 95,7% were aware of the syrup, and 69,8% were aware of both adult and paediatric formulations.
Perceived Efficacy
The syrup was considered useful for reducing cough by 93,9% of respondents, with 77,2% rating it as very or extremely effective.
Regarding cough improvement, 82% reported a moderate or significant improvement, while only 3,4% perceived no change.
For throat discomfort relief, 93,1% gave a positive assessment, with 44,4% rating it extremely effective.
In mucus expulsion, 91,5% found it effective, and 88,1% rated it positively for overall relief of respiratory symptoms (Figure 4).
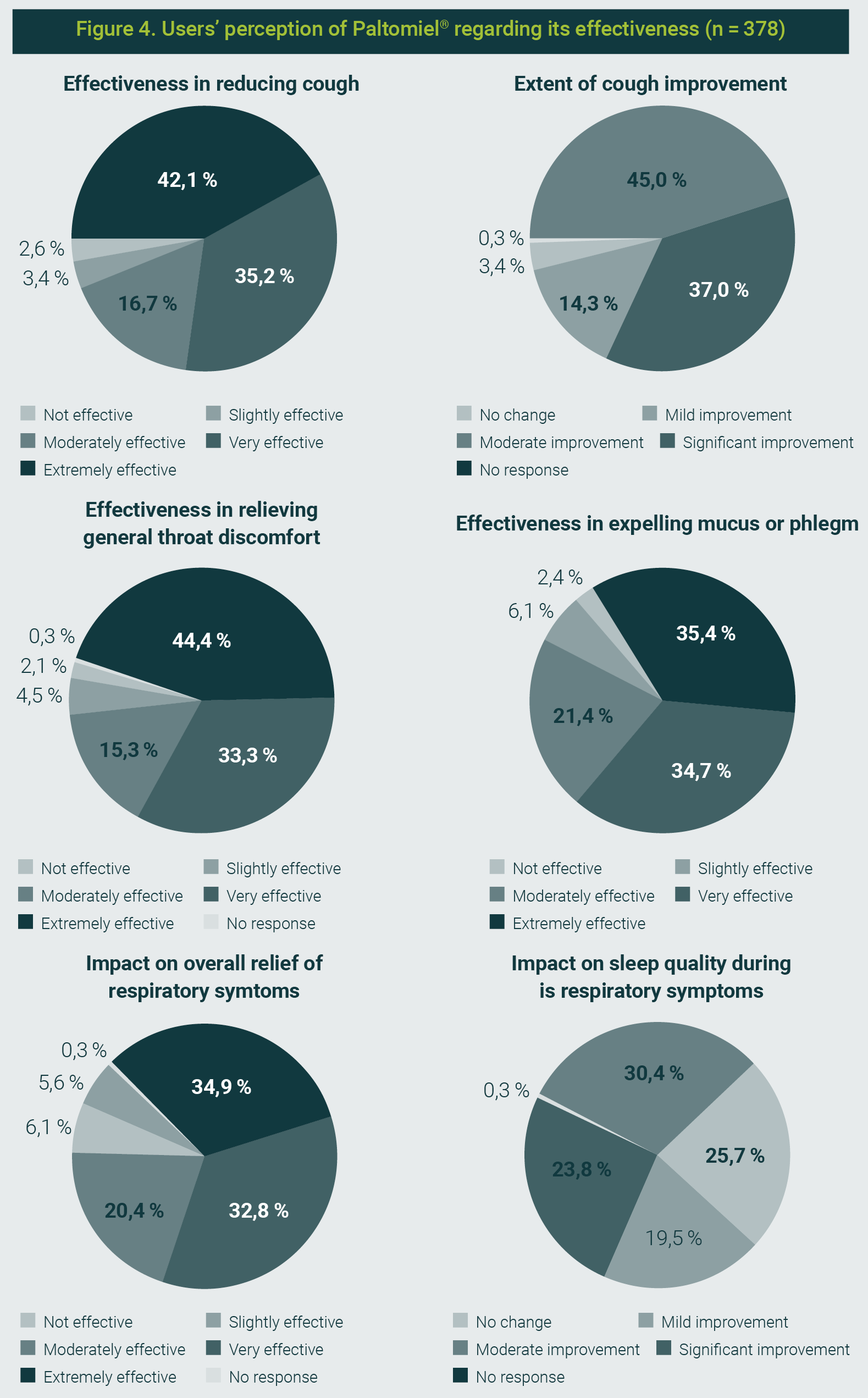
The figure is property of the authors
Adverse Events and Tolerability
The findings were the following:
- 1,3% reported any adverse reaction,
- 97,9% indicated no side effects,
- 54,2% reported improved sleep quality during respiratory illness episodes when using the syrup.
Overall Satisfaction
Satisfaction was high, with 80,4% describing themselves as very or extremely satisfied and only 2,1% expressing dissatisfaction.
Open-ended comments frequently praised the syrup’s effectiveness, natural composition, and regular use within families, though some noted excessive sweetness or greater benefit in early stages of cough.
In both the Chilean and international markets, there is a wide range of standardized natural pharmaceutical products and mixed formulations marketed for various pathologies, either as first-line treatments or as adjuvants. Many of these products lack robust scientific support or clinical studies to substantiate their efficacy and/or safety. Nonetheless, they are widely used and highly valued by the public due to long-standing traditional use.
For such products, it is essential to compile supporting scientific evidence, obtain expert-opinion certifications, conduct perception studies, and, where feasible, conduct at least limited clinical trials to strengthen confidence in their therapeutic role.
In Chile, most herbal cough syrups are based on Hedera helix (ivy).5 Paltomiel® combines several natural active ingredients, each with documented efficacy, although no specific clinical trials have evaluated the exact formulation.
The natural components in the studied formulation have traditionally been used for respiratory conditions due to their anti-inflammatory, expectorant, antitussive, and antispasmodic properties:
- Eucalyptus globulus contains eucalyptol (1,8-cineole), which has mucolytic, anti-inflammatory, and bronchodilatory effects that improve lung function and relieve cough.37,38
- Pimpinella anisum provides carminative, antioxidant, and antitussive effects that can help reduce bronchial irritation.39,40
- Persea americana contributes flavonoids with anti-inflammatory and antioxidant activity, although it has been less extensively studied.
- Honey enhances therapeutic action through antimicrobial and demulcent properties, reducing night-time cough and improving sleep.41
- Hedera helix, although not an ingredient in Paltomiel®, is referenced for comparison, as its triterpenic saponins act as mucolytics and bronchodilators.7
Survey findings strongly support these properties, indicating high levels of awareness and acceptance among physicians: 96,5% were familiar with the product, and more than 80% had prescribed it.
Effectiveness was remarkably well rated for productive cough (80%) and to a slightly lesser extent for dry cough (75%).
Organoleptic tolerability was judged good or excellent by 81,4%, and adverse events were rare.
Willingness to continue prescribing was also high (80%).
The main barrier to broader recommendation was the absence of formulation-specific clinical evidence, which aligns with the recognised need for formal validation of phytotherapeutic combinations.
From the users’ perspective, perceptions were similarly positive. Almost all knew the product, and over 93% considered it effective against cough. It was also valued for relieving throat discomfort (93,1%), aiding mucus clearance (91,5%), and improving general respiratory symptoms (88,1%). Notably, 54,2% reported improved sleep quality during illness episodes, highlighting honey's demulcent properties. Adverse effects were minimal, with 97,9% reporting none.
These findings position the formulation as a well-tolerated, widely accepted phytotherapeutic alternative for productive cough and related respiratory symptoms. However, the absence of randomised controlled trials (RCTs) means that its efficacy cannot yet be quantitatively compared with other available treatments, such as Hedera helix-based syrups or synthetic expectorants.
Conducting RCTs across Phases I-IV would allow rigorous evaluation of its safety, efficacy, and comparative effectiveness.
In addition, given its anti-inflammatory and expectorant properties, future research could explore its role as a complementary treatment for viral respiratory infections, including post-viral cough syndromes and post-COVID-19 bronchial inflammation, in which persistent cough and airway irritation are common.42,43
Overall, the evidence gathered in this study suggests that Paltomiel®, as an example of a mixed formulation of several complementary or synergistic natural extracts for cough, is a credible, well-received national phytotherapeutic product, with strong endorsement from users and physicians. Strengthening its scientific basis through clinical trials would not only reinforce its therapeutic position but also facilitate broader clinical adoption.
The authors sincerely thank the physicians and users who voluntarily participated and completed the surveys, making this study possible.
The authors declare that they have no conflicts of interest.
| 1. | Pérez BF, Méndez GA, Lagos RA, Vargas MSL. Dinámica y patología del barrido mucociliar como mecanismo defensivo del pulmón y alternativas farmacológicas de tratamiento. Rev Med Chil. 2014;142(5):606-15. |
| 2. | Fanta CH. Tos. En: Loscalzo J, Fauci A, Kasper D, Hauser S, Longo D, Jameson J (editores). Harrison. Principios de Medicina Interna. 21a ed. McGraw-Hill Education; 2022. |
| 3. | Chang CC, Cheng AC, Chang AB. Over-the-counter medications to reduce cough as an adjunct to antibiotics for acute pneumonia in children and adults. Cochrane Database Syst Rev. 2014;2014(3):CD006088. |
| 4. | Manti S, Tosca MA, Licari A, Brambilla I, Foiadelli T, Ciprandi G, et al. Cough remedies for children and adolescents: Current and future perspectives. Paediatr Drugs. 2020;22(6):617-34. |
| 5. | Sierocinski E, Holzinger F, Chenot JF. Ivy leaf (Hedera helix) for acute upper respiratory tract infections: An updated systematic review. Eur J Clin Pharmacol. 2021;77(8):1113-22. |
| 6. | Her L, Kanjanasilp J, Chaiyakunapruk N, Sawangjit R. Efficacy and safety of eucalyptus for relieving cough: A systematic review and meta-analysis of randomized controlled trials. J Integr Complement Med. 2022;28(3):218-26. |
| 7. | Mashat GD, Hazique M, Khan KI, Ramesh P, Kanagalingam S, Ul Haq Z, et al. Comparing the effectiveness of honey consumption with anti-cough medication in pediatric patients: A systematic review. Cureus. 2022;14(9):e29346. |
| 8. | Dezube. Mecanismos de defensa del sistema respiratorio. [Consultado el 3 de febrero, 2026]. Disponible en URL: https://www.msdmanuals.com/es/hogar/trastornos-del-pulmón-y-las-vías-respiratorias/biología-de-los-pulmones-y-de-las-vías-respiratorias/mecanismos-de-defensa-del-sistema-respiratorio |
| 9. | Nadler PL, Gonzales R. Tos. En: Papadakis MA, McPhee SJ, Rabow MW, McQuaid KR (editores). Diagnóstico clínico y tratamiento 2024. McGraw Hill; 2024. |
| 10. | Sucher B. Tos y otros síntomas respiratorios inespecíficos. En: DiPiro JT, Yee GC, Posey L, Haines ST, Nolin TD, Ellingrod V (editores). Farmacoterapia: un enfoque fisiopatológico. 11a ed. McGraw-Hill Education; 2020. |
| 11. | Cherniack RM, Cherniack L. The cough reflex. In: Respiration in Health and Disease. 3rd ed. Philadelphia: WB Saunders; 1983. |
| 12. | Chesnutt MS, Prendergast TJ. Enfermedad pulmonar. En: Hammer GD, McPhee SJ (editores). Fisiopatología de la enfermedad: Introducción a la medicina clínica. 7a ed. New York: McGraw-Hill; 2013. |
| 13. | Bonvini SJ, Birrell MA, Smith JA, Belvisi MG. Targeting TRP channels for chronic cough: From bench to bedside. Naunyn-Schmiedeb Arch Pharmacol. 2015;388(4):401-20. |
| 14. | Canning BJ, Chang AB, Bolser DC, Smith JA, Mazzone SB, McGarvey L, et al. Anatomy and neurophysiology of cough: CHEST guideline and expert panel report. Chest. 2014;146(6):1633-48. |
| 15. | Chang AB. The physiology of cough. Paediatr Respir Rev. 2006;7(1):2-8. |
| 16. | Rodríguez A, Izuibejeres C, González V. Cough suppressants and synthetic expectorants. Arch Pediatr Urug. 2021;92(Suppl 2). |
| 17. | MD Anderson Cancer Center Madrid. Antitusígeno. 2025 [Retrieved on February 4th, 2026]. Available from URL: https://mdandersonhospiten.es/elcancer/glosario/antitusigeno |
| 18. | Manayi A, Vazirian M, Saeidnia S. Echinacea purpurea: Pharmacology, phytochemistry and analysis methods. Pharmacogn Rev. 2015;9(17):63-72. |
| 19. | Medeiros Peixoto D, Rizzo JA, Schor D, Silva AR, Cavalcanti de Oliveira D, Solé D, et al. Use of honey associated with Ananas comosus(Bromelin) in the treatment of acute irritative cough. Rev Paul Pediatr. 2016;34(4):412-7. |
| 20. | Bera K, Nosalova G, Sivova V, Ray B. Structural elements and cough suppressing activity of polysaccharides from Zingiber officinale rhizome. Phytother Res. 2016;30(1):105-11. |
| 21. | Mahboubi M. Marsh mallow (Althaea officinalis L.) and its potency in the treatment of cough. Complement Med Res. 2020;27(3):174-83. |
| 22. | Völp A, Schmitz J, Bulitta M, Raskopf E, Acikel C, Mösges R. Ivy leaves extract EA 575 in the treatment of cough during acute respiratory tract infections: Meta-analysis of double-blind, randomized, placebo-controlled trials. Sci Rep. 2022;12(1):20041. |
| 23. | Harris D. Quercetin, mast cell activation syndrome, and histamine intolerance. 2024 [Retrieved on February 4th, 2026]. Available from URL: https://www.eds.clinic/articles/quercetin-mast-cell-activation-syndrome-and-histamine-intolerance |
| 24. | Arooj B, Asghar S, Saleem M, Khalid SH, Asif M, Chohan T, et al. Anti-inflammatory mechanisms of eucalyptol-rich Eucalyptus globulus essential oil alone and in combination with flurbiprofen. Inflammopharmacology. 2023;31(4):1849-62. . |
| 25. | Pahlavani N, Malekahmadi M, Firouzi S, Rostami D, Sedaghat A, Moghaddam AB, et al. Molecular and cellular mechanisms of the effects of Propolis in inflammation, oxidative stress and glycemic control in chronic diseases. Nutr Metab (Lond). 2020;17:65. |
| 26. | Ożarowski M, Karpiński TM. The effects of Propolis on viral respiratory diseases. Molecules. 2023;28(1):359. |
| 27. | Pourova J, Dias P, Pour M, Bittner Fialová S, Czigle S, Nagy M, et al. Proposed mechanisms of action of herbal drugs and their biologically active constituents in the treatment of coughs: An overview. PeerJ. 2023;11:e16096. |
| 28. | MedicalNewsToday. Chen H. How to prevent a cold. 2023 [Retrieved on February 4th, 2026]. Available from URL: https://www.medicalnewstoday.com/articles/322394 |
| 29. | Centro Médico Imbanaco. Salgado TE. Tos, 2025. [Retrieved on February 4th, 2026]. Available from URL: http://www.carlosenriquesalgado.com/index.php/problemas-clinicos-frecuentes/tos |
| 30. | PAM. Expectorantes, antitusivos y mucolíticos. Manual de Formación Continuada. 2022 [Retrieved on February 4th, 2026]. Available from URL: https://www.farmaceuticos.com/pam/temas/formacion-continuada/expectorantes-antitusivos-y-mucoliticos/ |
| 31. | Cortes-Telles A, Che-Morales JL, Ortíz-Farías DL. Estrategias actuales en el manejo de las secreciones traqueobronquiales. Neumol Cir Torax. 2019;78(3):313-323. |
| 32. | NIH. Instituto Nacional del Cáncer. Broncodilatador. 2025 [Retrieved on February 4th, 2026]. Available from URL: https://www.cancer.gov/espanol/publicaciones/diccionarios/diccionario-cancer/def/broncodilatador |
| 33. | Knop Laboratorios. Paltomiel: folleto de información al profesional. 2017. |
| 34. | Superintendencia de Salud. Departamento de Estudios y Desarrollo. Documento de trabajo. Caracterización de las especialidades médicas en Chile en 2022. [Retrieved on February 4th, 2026]. Available from URL: https://www.superdesalud.gob.cl/app/uploads/2024/02/articles-26547_recurso_1.pdf |
| 35. | Atkinson MJ, Sinha A, Hass SL, Colman SS, Kumar RN, Brod M, et al. Validation of a general measure of treatment satisfaction, the Treatment Satisfaction Questionnaire for Medication (TSQM), using a national panel study of chronic disease. Health Qual Life Outcomes. 2004;2:12. |
| 36. | Vernon MK, Revicki DA, Awad AG, Dirani R, Panish J, Canuso CM, et al. Psychometric evaluation of the Medication Satisfaction Questionnaire (MSQ) to assess satisfaction with antipsychotic medication among schizophrenia patients. Schizophr Res. 2010;118(1-3):271-8. |
| 37. | Dhakad AK, Pandey VV, Beg S, Rawat JM, Singh A. Biological, medicinal and toxicological significance of eucalyptus leaf essential oil: A review. J Sci Food Agric. 2018;98(3):833-48. |
| 38. | Juergens LJ, Worth H, Juergens UR. New perspectives for mucolytic, anti-inflammatory and adjunctive therapy with 1,8-cineole in COPD and asthma: Review on the new therapeutic approach. Adv Ther. 2020;37(5):1737-53. |
| 39. | Gulçin İ, Oktay M, Kıreçcı E, Küfrevıoǧlu Öİ. Screening of antioxidant and antimicrobial activities of anise (Pimpinella anisum L.) seed extracts. Food Chem. 2003;83(3):371-82. |
| 40. | Samojlik I, Mijatović V, Petković S, Skrbic B, Božin B. The influence of essential oil of aniseed (Pimpinella anisum L.) on drug effects on the central nervous system. Fitoterapia. 2012;83(8):1466-73. |
| 41. | Cohen HA, Rozen J, Kristal H, Laks Y, Berkovitch M, Uziel Y, et al. Effect of honey on nocturnal cough and sleep quality: A double-blind, randomized, placebo-controlled study. Pediatrics. 2012;130(3):465-71. |
| 42. | López-Cortés A, Guerrero S, Ortiz-Prado E, Yumiceba V, Vera-Guapi A, León-Cáceres A, et al. Pulmonary inflammatory response in lethal COVID-19 reveals potential therapeutic targets and drugs in phases III/IV clinical trials. Front Pharmacol. 2022;13:833174. |
| 43. | Sanhueza S, Vidal MA, Hernandez MA, Henriquez-Beltran ME, Cabrera C, Quiroga R, et al. Clinical and pulmonary function analysis in long-COVID revealed that long-term pulmonary dysfunction is associated with vascular inflammation pathways and metabolic syndrome. Front Med (Lausanne). 2023;10:1271863. |
All Rights Reserved® 2019
Latin American Journal of Clinical Sciences and Medical Technology,Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.
All Rights Reserved® 2019
Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.