| ** | Latin American Journal of Clinical Sciences and Medical Technology is an open access magazine. To read all published articles and materials you just need to register Registration is free of charge. Register now If you already have registered please Log In | ** |
aDirección de Farmacovigilancia, Asociación Farmacéutica Mexicana, Ciudad de México, México; bDirección General, Hospital General de Cuautla, Morelos, México; cInosan Biopharma, S.A. Ciudad de México.
Corresponding Author: , . Telephone number: ; e-mail: raulsoria2000@yahoo.com.mx
Lat Am J Clin Sci Med Technol. 2019 Apr;1:7-20.
Received: February 5th, 2019.
Accepted: March 28th, 2019.
Published: April 23rd, 2019.
Views: 8153
Downloads: 18
Antivenoms (AVs) are biological therapies indicated in the treatment of envenoming by bites or stings of venomous animals. However, access, supply problems, quality of manufacturing, sociocultural issues, and regulatory aspects are factors related to the effectiveness and safety of AVs. Likewise, the controlled clinical research is limited apparently for the low prevalence registered in tropical regions such as Latin America, Middle East, Africa, and Asia; therefore, a lack of evidence may elicit an unsuitable approach on the emergency or intensive care units. Moreover, the clinical trials are controversial because they would not reflect the reality inside the clinical setting of envenoming. Accordingly, there are factors influencing the therapeutic response to AVs. Here, the implementation of pharmacovigilance (PV) may help to improve the management of the AVs-related risks through the identification and assessment of suspected drug adverse reactions, ineffectiveness, and other drug-related problems. Notwithstanding, the knowledge of the PV within clinical toxicology, emergency, and intensive care units is limited. Then, the PV must be established as a routine activity within an integral process of the medical attention of envenoming by bites or stings from venomous animals (BSVA) to guarantee a suitable and safe medication. The aim of this paper is outlining an overview about the clinical setting of envenoming, the current treatment and its relationship with the PV as a complementary activity to improve the quality, effectiveness, safety of AVs, through a perspective of experts in Mexico.
Los antivenenos (AV) son terapias biológicas indicadas para el tratamiento de envenenamiento por mordeduras o picadura de animales venenosos. El acceso, suministro, calidad de fabricación, asuntos socioculturales y regulatorios son aspectos que se relacionan con la efectividad y seguridad de los AV. Igualmente, los estudios clínicos controlados son escasos debido, aparentemente, a la baja incidencia en regiones tropicales, tales como América Latina, Medio Oriente, África y Asia; por tanto, la falta de evidencia puede propiciar un abordaje inadecuado en urgencias o en las unidades de cuidados intensivos. Además, los estudios clínicos despiertan controversia porque no reflejan la realidad en el medio clínico del envenenamiento. Todo ello incide en la respuesta terapéutica a losAV. La implementación de la farmacovigilancia (FV) podría contribuir a mejorar el manejo de riesgos relacionados con los AV mediante la identificación y evaluación de sospechas de reacciones adversas, falta de eficacia y otros problemas relacionados con la medicación. No obstante, el conocimiento de la FV en toxicología clínica, emergencias y unidades de cuidados intensivos es limitado. Por tanto, la FV debe establecerse como una práctica de rutina en el proceso integral de atención médica de envenenamiento por mordeduras o piquetes de animales venenosos para garantizar una medicación adecuada y segura. El objetivo de este documento es describir un panorama del ambiente clínico del envenenamiento, el tratamiento actual y su relación con la FV, como una actividad complementaria para mejorar la calidad de vida efectividad y seguridad de los AV mediante una perspectiva de expertos en México.
The antivenoms (AVs) are biological drugs defined by World Health Organization (WHO) as the purified fraction of immunoglobulins (Ig) or immunoglobulin fragments fractionated —Fab or F(ab’) 2— from the plasma of animals that have been immunized against one or more venoms; hence, their suitable use can prevent or reverse most of the negative effects of envenoming after the administration of an intravenous infusion of the AV.1 However, bites or stings from venomous animals (BSVA) may happen in remote areas, quite far from a specialized hospital (i.e., in rural areas) or urban areas (i.e., by occupational exposure to venomous animals). Accordingly, primary attention will be provided, at best, on small health centers which not necessarily have AVs available for an emergency case. Therefore, the patients are referred to another specialized hospital depending mainly on its facilities and on the local health system.2,3
These circumstances should be considered because medical attention timing is a very important prognosis factor to ensure efficacy and safety of the treatment. If the hospitals had biological therapies (BT) in emergency or intensive care units (ICU), there would be a problem: they often do not have a suitable pharmacovigilance (PV) process to guarantee the effectiveness, safety and rational BT use. Thus, it is necessary to establish a PV method inside hospitals in order to support the prognosis of a critical patient during the medical intervention and follow-up.
However, AVs may not have the same importance for health programs like other diseases and BT. Unfortunately, in emerging regions such as Latin America and Africa, there are not sufficient reimbursement programs to encourage the clinical research to develop new treatments. Accordingly, lack of access to AVs is common because it is considered a low-prevalence disease in many countries.
Therefore, the role of academies of medicine, toxicology, pharmacology, pharmacy, as well as the civil society must support the establishment of a unified knowledge, national registry, better clinical trials, continuous medical update, diffusion of training programs, access, and governmental awareness about envenoming by BSVA for the benefit of patient safety. WHO has recommended to establish public health strategies as an urgent measure to ensure the availability of safe, effective, and affordable AVs, especially for developing countries (i.e. in Mexico), as well as to improve the regulatory control over the manufacture, import and sale.1 Therefore, a complementary tool to achieve this objective is PV.
PV is defined by the WHO as “the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem”.4
Currently, 194 countries are part of WHO and 131 countries are members of Program for International Drug Monitoring (PIDM) coordinated by the Uppsala Monitoring Center (UMC). Mexico is part of PIDM since 1999, represented by the PV National Center of the Federal Commission for the Protection against Sanitary Risk in the Health Ministry (COFEPRIS). Mexican PV is regulated by Mexican Official Standard NOM-220-SSA1-20165, which is consistent with the guidelines from the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use, specifically, ICHE2E.6 Although technical requirements for drug registration are recommended here, they are mainly focused on compliance of regulatory authorities and pharmaceutical industry. Thus, AVs have limited clinical data because the local guidelines are focused on the compliance of reports (i.e. coming from clinical trials), but they do not make a reference on the attention of critical patient where the PV application is different from others medical areas, especially in the attention of vulnerable groups such as geriatric and pediatric patients.4,7,8 Therefore, it is necessary the implementation of PV in the emergency unit and ICU for cases of envenoming by BSVA to distinguish the adequately effectiveness and safety profile of AVs.
The aim of this paper is outlining an overview about the clinical setting of envenoming, the current treatment and its relationship with the PV as a complementary activity to improve the quality, effectiveness, safety of AVs, through a perspective of experts in Mexico.
Envenoming by BSVA is a process whereby the venom is injected in a patient through the bite or sting of a venomous animal or ingestion of toxins produced by animals or plants.9 This perspective article will be focused on three envenoming of medical importance: 1) scorpion sting, 2) spider bites, and 3) snake bite.
Epidemiology
Cases of envenoming by endemic species have been reported in tropical regions of countries such as Mexico, United States, Brazil, Colombia, Costa Rica, Ecuador, Venezuela, Argentina, Australia and specifically in Trinidad and Tobago, Turkey, Sri Lanka, and Nigeria. Global epidemiology data has been suggested around 3 million cases per year with more than 150,000 deaths by BSVA.10,11
Besides being not easily available, these data could be underestimated because not all patients receive quick attention at the hospital, mainly due to other causes such as cultural belief, lack of infrastructure/national registries, non-updated/incomplete data or inaccessibility to treatment.1 Because of its apparent low prevalence, envenoming by BSVA could be classified as orphan disease only if it meets some regulatory requirements.12 More epidemiological data are needed for national prevention programs and registry of cases in order to improve the health attention, especially in vulnerable population.
In particular, the Epidemiology General Direction from Mexico reported 336 268 (100%) new cases of BSVA in 2017 mainly due to scorpion stings, 295 321 (87.8%) of them were confirmed cases. Likewise, there were 37 213 (11%) accidents including spider species bites and 3 734 (1.1%) cases of snake bites.
Apparently, BSVA with major prevalence in Mexico were scorpion species stings. They ranked number 13 as primary cause of disease at national level in 2015 (rate of 0.03% per 100 000 inhabitants), and the third place on national mortality over children between 1-4 years old with a mortality rate of 0.2% per 100 000 inhabitants (national mortality by subjected causes to epidemiological surveillance). They were only preceded by deaths due to acute respiratory infection and acute diarrheal disease.
The Mexican states with major number of reported cases are Jalisco, Guanajuato, Guerrero, Morelos, Michoacán, Nayarit, Puebla, Colima, Estado de México, Durango, Sinaloa, Sonora and Zacatecas. The more affected populations were adults, young, and children.13
Scorpion stings
More than 1.2 million of cases per year, leading to 3 250 deaths worldwide, have been estimated due to scorpion stings. Dangerous scorpion genus/species for human have been described in Latin America such as Centruroides sp (i.e. Bark scorpion —Centruroides sculpturatus—) and Tityus sp (i. e. Brazilian yellow scorpion —Tytus serrulatus—), among others.14,15
Spider bites
Unfortunately, there is not a global appraisal and reports are confusing because of methodological deficiencies and a lack of differential diagnosis.
Most spider species are not considered dangerous; however, Toxic Exposure Surveillance System from American Association of Poison Control Centers reported a total 48 411 cases in 2016. Only 15 480 cases were treated into Health Care Facility which they confirmed 7 deaths. Likewise, between 2001 and 2010, an estimated of 169 010 cases were attended in the emergency unit by bite and sting from insects or arachnids in United States. Arachnids were responsible for 20.8% of cases, the spider species were responsible for 2/3 of all cases, but only the 2.2% resulted in envenoming cases by spider bite.16,17
Spider genus / species dangerous for human have been described in Latin America such as Latrodectus sp (i.e. black widow —Latrodectus mactans—) and Loxocele sp (i.e. violinist, brown or recluse spider —Loxosceles laeta—), among others.18,19
Snake bites
This type kills >100 000 people and maims >400 000 people every year. Snake genus/species have been described in Latin America such as Crotalus sp (i.e. Mexican west coast rattlesnake —Crotalus basiliscus—) and Micrurus sp (i. e. aquatic coral snake —Micrurus surinamensis—), among others.20,21
Summary of signs and symptoms
Venom is composed by a complex mixture of non-protein and protein substances (i.e. metallic ions, lipids, glycosides, free amino acids), but especially hydrolytic and proteolytic enzymes as well as peptide toxins or non-toxic peptides (depending on species).22,23
Table 1 shows a comparison between several signs and symptoms from human life-threatening envenoming animals.
| Table 1. Comparison between several signs and symptoms from human life-threatening BSPA. | ||||
|---|---|---|---|---|
| Envenoming type | Family* | Several representatives genus of medical importance** | Toxicity type*** | Clinical manifestations**+ |
| Scorpion sting14, 24, 25 | Buthidae. | Centruroides sp., Tityus sp., Hemiscorpius sp., Odonthobuthus sp., Cercophonius sp., Parabuthus sp., Leiurus sp., Lychas sp., Isometrus sp., esobuthus sp., Hottentotta sp., Urodacus sp., Androctonus sp. and Buthus sp. | Neurotoxic, cardiotoxic, myotoxic, cytotoxic. | Paralysis, nystagmus, paresthesia, muscle weakness, burning pain, salivation and lachrymation, respiratory distress syndrome, sweating, tachypnea, coma, convulsions, mydriasis, erythema, cardiovascular disorders, nausea, vomiting, gastric distention, pulmonary edema, cardiac dysfunction, impaired hemostasis, muscle spasm or fascisculation, pancreatitis, and multiple organ failure. |
| Spider bite24, 26, 27 | Hexathelidae, Sicariidae, Ctenidae, Theridiidae, Lycosidae, Dipluridae. | Latrodectus sp., Loxocele sp., Lampona sp., Achaearanea sp., Sphodros sp., Phoneutria sp., Badumna sp., Atrax sp., Hadronyche sp., Cheiracanrhium sp., Trechona sp., Microhexura sp., Tegenaria sp., Brachythele sp., and Evagrus sp. | Neurotoxic, necrotoxic, cardiotoxic, nephrotoxic. | Local pain, late erythema, edema, ataxia, coagulopathy, headache, lethargy, irritability, myalgia, tremor, fasciculation, mottled dark or hemorrhagic foci, with or without local blistering, skin necrosis, renal failure, hemolysis, and thrombocytopenia. |
| Snake bite20, 24, 28 | Viperidae, Elapidae, Hydophiidae. | Crotalus sp., Vipera sp., Sistrutus sp., Micruroides sp., Dendroaspis sp., Naja sp., Micrurus sp., Hydrophis sp., Oxyuranus sp., Ophidiophagus sp., Bothrops sp., Acanthophis sp., Agkistrodon sp., Dendroaspis sp., Bungarus sp., Bitis sp., Laticauda sp., Pseudonaja sp., Timeresurus sp., Lachesis sp., and Notechis sp., | Hemotoxic, neurotoxic, nephrotoxic, cardiotoxic, myotoxic, cytotoxic. | Coagulopathy, hemorrhages, Thrombocytopenia, cardiac arrest, blurred vision, nausea, vomiting, stroke, muscle paralysis, pain, weakness, espiratory distress, acute kidney injury, swelling, ecchymosis, hypotension and altered mental status. |
| Abbreviations: sp.: species. * Hazardous for human life. There are numerous genders, species or endemic ariants which will not be fully mentioned here. ** They are not organized by family or taxonomy. They are all examples. *** Mainly, but it may vary between genus, specie or variants of them. Underlined emphasis is predominant toxicity. | ||||
Hence, the clinical manifestations are heterogeneous and they will depend on the animal type, bite or sting type, amount of injected venom, place of depot, attacked body surface, age of patients and their health condition.29 Understanding of natural history of disease will support the challenges from addressing for diagnosis, severity, treatment and prognosis. BSVA may cause other potentially serious complications for the patient, such as infections, amputations, compartmental syndrome, recurrences, scar deformations, and psychological traumas. The likelihood of death must be low if there is an available treatment in the hospital and if clinical approach is suitable.10,30 Therefore, attention for envenoming must be comprehensive and multidisciplinary.
Systemic response to venom
Immune response to snake and scorpion venoms has been described previously.31-33 The patients could develop similar signs and symptoms of allergy before administration of AV. Currently, anaphylaxis rare cases (≤1%) by snake bites have been documented, but the differential diagnosis is not clear yet.34
Health professionals must consider that not only the specific antibodies against the venom are produced as immune response after envenoming (i.e. IgE or IgG), but also they could induce the production of others mediator types such as Na+, Ca+2, K+, adrenaline, aldosterone, nitric oxide, angiotensin converting enzyme, and angiotensin II. They may trigger several additional clinical manifestations to envenoming.35
Compartment syndrome
It is an uncommon neuromuscular complication after a BSVA. It is the result of an edema (i.e. by snake bite) causing an increase in pressure within one or more fascial compartments, hypoxia and acidosis, which further increases capillary permeability and interstitial liquid extravasation.24 This may impair blood flow and irreversibly damage the muscle and nerve with a potential loss of functionality. Early diagnosis during admission is crucial to reduce this secondary complication.36
Diagnosis and severity
Consensus over envenoming diagnosis by BSVA is limited because the diversity of venomous animals is very wide; thus, the clinical manifestations and severity classification will depend on complex variables both entomology/herpetology (as public health system) and clinical practice in each region. Then, defining a unique criteria for the envenoming approach is a challenge. However, there are evidence-based recommendations which may support the diagnosis and prognosis. Table 2 shows a summary of recommendations over diagnosis, classification, and laboratory tests to surveillance from envenoming evolution by BSVA. Unfortunately, there is not a consensus about the approach for spider bites. Conversely, the local guidelines and medical reviews for a better understanding about diagnosis and treatment should be noted.29,37-41
| Table 2. Summary of recommendations over diagnosis, classification, treatment, and laboratory tests to the surveillance from envenoming by BSVA. | |||
|---|---|---|---|
| Envenoming type | Grade* | Grade/Start AV+CT/Vials** | Laboratory test |
| Scorpion sting25, 29, 42, 43 | I. Localized symptoms. II. General and systemic symptoms. III. Life-threatening prognosis. | I. No + emergency medical assistance + acetylsalicylic acid 10 mg/kg orally every 4 h. II. Yes + idem + prazosin 30 μg/kg orally every 6 h + midazolam 0.05-0.2 mg/kg orally or intravenously (or diazepam 0.5 mg/kg intravenously or rectally every 12 h). Precaution: There is a risk of respiratory depression with CT. III. Yes + idem + transfer to ICU. Number of vials: limited to 1-5 | Basic tests, platelet count, ABG, CPK, urinalysis and cardiac enzymes and cardiovascular tests. Evidence of local infection requires standard microbiologic culture and sensitivity testing to start suitable antimicrobial therapy. |
| Spider bite19, 29, 44 | NA or well, generally is mild, moderate and severe. | NA. + emergency medical assistance. The tetanus vaccine (Clostridium tetani) is recommended. If necessary, transfer to ICU. Number of vials: limited to 1-3 + dapsone 1 mg/kg/day divided on 2 days during 10 days. | Basic tests, platelet count, ABG, CPK, urinalysis, cardiac enzymes. Evidence of local infection requires standard microbiologic culture and sensitivity testing to start suitable antimicrobial therapy. |
| Snake bite29, 40, 45 | 0=non-envenomation (dry bite) I. Local tissue effects (mild). II. Hematologic venom effects (moderate). III. System venom effects (severe). or IV. Life-threatening | 0. No + emergency medical assistance I. No + idem + analgesic and antihistaminic drugs. A tetanus vaccine (Clostridium tetani) is recommended. II. Yes + idem + opioids are preferred over NSAIDs because of bleeding risk (coagulopathy or thrombocytopenia). Premedication (i.e. steroids) and decreased infusion velocity (faster→slower) to avoid drug hypersensitivity. It has not shown significant benefits. III. Yes + idem + transfer to ICU Number of vials: Frequently between 4-6. It has been recommended until +20 vials by patient, but this may increase the risk of ADR or serum sickness. | Basic tests, PT/INR, APTT, fibrinogen (measure, not calculated) D-dimer, platelet count, ABG, CPK, urinalysis, cardiac enzymes. Evidence of local infection requires standard microbiologic culture and sensitivity testing to start a suitable antimicrobial therapy. |
| NA: not available, CT: concomitant treatment, ABG: arterial blood gas, CPK: creatinine phosphokinase, APTT: activated partial thromboplastin time, INR: international normalized ratio, PT: prothrombin time, ICU: intensive care unit. *Classification made by consensus in spite of Delphi method was modified. There is not a classification to spider bite. **Low doses → High doses depending of envenoming grade, clinical evolution and venomous animal species. Concomitant treatment was recommended by evidence-based criteria. | |||
Starting treatment
Before starting treatment, the differential diagnosis must be based on physical examination and on clinical record. Due to specific circumstances in clinical setting, it will not always be possible to identify the venomous specie regarding all entomology/herpetology criteria. Accordingly, the criteria for starting treatment will be based on human life-threatening signs and symptoms, taking into consideration that not all BSVA induce envenoming. For instance, 20-25% patients suffer “dry bite” of snake, that is to say, venom is not released on damaged site, but if patients develop any or minimum evidence of envenomation they should be given antivenom.46
Types of AV
AVs are neutralizing proteins with or without changes over chemical structure for improving their affinity-avidity, pharmacokinetics (PK) or pharmacodynamics (PD) properties. They are biochemically called glycoproteins or immunoglobulins G (IgG) obtained by purification from hyperimmunized serum of animals (i. e. horses).47
Nowadays, three types of AV have been developed: 1) polyvalent IgG, 2) polyvalent fragments of IgG —F(ab’)2—, and 3) polyvalent fragments of IgG —Fab—. The two last ones were structurally modified by enzymatic digestion on key binding sites with the purpose of eliminating Fc portion of IgG associated with effector mechanisms such as activation of complement (i. e. C1q), proinflammatory cells (i. e. mast cells) or ADR (type 1 hypersensitivity), among others.48,49
Dosage
Doses scheme is still used empirically due to the lack of robust clinical trials. There are not comprehensive studies of posology, but the potency may determine the doses of AV required to neutralize the toxins. Greater therapeutic effect could be exerted with AVs of high potency and specificity. Nevertheless, there are not sufficient clinical trials “head-to-head” comparing the low potency AVs against high potency AVs. The non-envenomation should not be treated with AV (see Table 2).
Moderate (grade II) or severe cases (grade III) should be treated starting with escalating doses under manufacturer-indicated protocol (low→ high) until decreasing signs and symptoms of envenoming and preserving permanent surveillance during hospitalization. Table 2 also shows a summary of the experts’ recommendations over treatment with AVs. Apparently, the doses for risk groups such as children, pregnant women, and elderly are not different from the conventional scheme. Unfortunately, there are not comprehensive clinical trials to confirm it.
Recommendations by Mexican authorities
Clinical practice guidelines (with evidence-based methods) for diagnosis and treatment in Mexico recommended the next start doses: 1) scorpion sting, 1-6 vials 2) spider bite, one vial (possibly more vials will be used depending on clinical response), and 3) snake bite, 4-25 vials. The patient will be observed and if there is no clinical improvement, another dose will be administered equal or in accordance with the recommendations of the clinical practice guide.50-52
Clinical pharmacology
AVs are therapeutic proteins administered by intravenous (i.v) or other parenteral route. They are also distinguished by means of its pharmacological activity, measured as potency (Effective doses —ED50—) or mass units (mg) associated with the affinity-avidity ratio.53
Pharmacodynamics (PD) is similar to monoclonal antibodies (MAbs), that is to say, they have neutralizing activity against specific antigens over conformational or lineal epitopes. However, several AVs are polyclonal; thus, the selectivity and specificity is minor than MAbs. This may be necessary because the protein composition of venom is heterogeneous and the antigens (or epitopes) would be not unique by species, only related to them. Therefore, PD mechanism of the AVs is neutralizing over the binding site(s) from antigen(s) also called epitope(s) and it (they) would be composed of continuous or discontinuous amino acid sequences.54-56
AVs as well as MAbs have an Ig-based structure, but cannot induce active immunity or cells B-mediated immunological memory against the venom.57,58 Likewise, long time ago, some researchers indicated a mechanism of “artificial passive immunity” as acting manner of the AVs but only when they contain the IgG (antiserum) with the complete structure. Consequently, some regulatory agencies worldwide may classify them as vaccines or essential biologicals.59
Currently, the fragments from F(ab’)2 and Fab would not necessarily meet this rule due to the previous changes on its chemical structure and advances over the manufacturing.60 Then, on the one hand, it would a regulatory re-classification of AVs be suitable due to the current technologies.
It is important to highlight that the pharmacology of AV is different from conventional drugs (i. e. acetaminophen) in terms of absorption, distribution, metabolism, elimination and excretion, adverse drug reactions, immunogenicity, serum sickness, and dosage.
Absorption
Predominantly, AVs will be administered intravenously with infusion velocity recommended by manufacturer of the AV; hence, the bioavailability may theoretically achieve up to 100%. On the other hand, the absorption mechanism after subcutaneous and intramuscular administration is not completely understood and some evidence shows a contradictory therapeutic effect with this route, may be because of differences over the bioavailability.60-63
Distribution
AVs as Ig-based therapeutic proteins do not bind to plasma proteins and they are strictly dependent on their limited capacity to diffuse through cell membranes because the IgG have a high molecular weight. Therefore, the volume of distribution (Vd) is expected to be relatively small or approximately equal to volume of plasma, regardless of dose used.64 Additionally, Fc portion of the IgG plays a crucial role both in half-life time and in the crossover to the placenta barrier because all subclasses of IgG are transported through them by the neonatal receptor FcRn expressed into several tissues. It is expected certain traces of AV in fetus.65 It is worth saying, the fragments F(ab’)2 and Fab have a less molecular weight than cIgG because the Fc portion was previously digested by industrial processes. Theoretically, the IgG fragments would have a better penetration at damaged tissue and absent of crossover toward fetus. Notwithstanding, it has been not tested on comprehensive preclinical models.
Metabolism, elimination and excretion
AVs are neutralizing protein mixtures with a long-half life time dependent on molecular weight. They will not be metabolized by the classical via of cytochrome P450 (phase I) or conjugation (phase II). Metabolism of AV will be chiefly activated by unspecific catabolic proteases. This results in a higher half life time with cIgG than the Fab and F(ab’)2 fragments.
When AVs are formed by cIgG (molecular weight ≥150 kDa), a minimal glomerular filtration rate is expected; therefore, only small amounts could be detected in the urine. Some fragments of ≤ 60 kDa (i.e. Fab or F(ab’)2) will be filtered and excreted by the kidney, with an elimination rate approximately equal to the rate of glomerular filtration. However, the elimination of IgG is often a saturable and slow process; so, the clearance of AVs would decrease with dose. It will depend of the expression of therapeutic target. Hence, the clearance of Fab and F(ab’)2 fragments would be higher than cIgG, i.e., the time inside the body will be higher on cIgG than Fab and F(ab’)2 fragments.66,67
Adverse drug reactions
Adverse Drug Reactions (ADR) have been reported during the infusion protocol up to >50%, especially during the first 20 min, 2 h, 4 h, 8 h, and 24 h after administration. Syndrome of release of cytokines and hypersensitivity reactions are rare (<5%) and all these include symptoms such as rash, bronchospasm, nausea, vomiting, headache and fever or well, severe symptoms as anaphylaxis, hypotension and altered level of consciousness, among others.68-70 However, the signs and symptoms often may be misinterpreted with those related to the natural history of envenoming because there is not a complementary tool or sufficient clinical insight to distinguish them. Likewise, temporary cessation of AV will solve some mild reactions. In case of severe anaphylaxis, the World Allergy Organization recommends an initial administration of intramuscular adrenaline into the lateral thigh, 0.01 mg/kg to maximum of 0.3 mg. To unresponsive patients, they recommend an adrenaline infusion (0.5 ml/Kg/h, of 1 mg in 100 ml) and titration according to response. Monitoring, nebulized salbutamol for bronchospasm, and intravenous atropine for severe bradycardia are recommended. Premedication to decrease the potential ADR is controversial because the limited clinical trials are not concluding.
The type of ADR among AVs apparently do not differ, but their frequency/severity have been reported and may depend on the quality or purity of AV.71,72 The physiopathology mechanism of acute reactions is uncertain, but it may be the result of type I hypersensitivity or a combination of non-immunes, immunes mechanisms, although ADR may occur in those patients without previous exposure to animal proteins, i.e. coming from equines.73
Immunogenicity
The development of an immune response against AVs may negatively impact its pharmacokinetics. For instance, it may increase the clearance, modify the binding site to epitopes or alter the safety and efficacy profile. Human antibodies against AVs may be neutralizing or non-neutralizing depending on its effect over the pharmacological activity. If the AVs came from horse, then, the human antibodies “anti-antivenom” or “anti-drug” may be also called human “anti-horse” antibodies, which would be different from the human “anti-toxin” antibodies addressed against the protein composition of venom from venomous animals.
Immunogenicity of BT is an issue poorly understood; however, several researches indicate a crucial role of some trigger factors such as the molecular weight, BT type, drug target, glycosylation profile, pharmaceutical excipients, purity grade, treatment time, doses frequency, via of administration, dosage, concomitant drugs, age, comorbidities, genetic profile, among others.74,75 Unfortunately, it has been not comprehensively assessed with AV.
Serum sickness
It is rarely (<5% of cases) the result of an excess of foreign antigens that form circulating immune complex with different molecular weight for a certain period of time (i.e. until 20 days) after administration of AV coming from heterologous serum.
Immune system cannot recognize them and induces the delayed production of other human antibodies “anti-immunocomplex” which are deposited over organs (i.e., the kidney) and tissues raising a long-term damage. Signs and symptoms of serum sickness are confusing but could be defined as lethargy, headache, muscle/joint aches, and fever or similar to allergy, and infections until tissue damage.76
It occurs with the AVs formed by cIgG (anti-serum), but the risk of immunocomplex formation should be lower with the fragments Fab and F(ab’)2 because the time into body is lower than cIgG and the Fc portion has been deleted, which is associated to secondary binding site and pro-inflammatory mechanisms.77 However, there are not robust clinical trials assessing it.
Recurrent signs
They are signs of envenomation occurring after stabilization of laboratory parameters, even with medication. They may happen during 24 hours or up to 2 weeks after the critical stage of envenoming. This phenomenon has been documented in the coagulopathy by snake bites78 but not with scorpion stings or spider bites.
Recurrent mechanism by snake bite is unclear, but it has been previously hypothesized that depots of non-neutralized venom may continue being released into the blood flow after antivenoms levels fall, which causes recurrent coagulopathy.79 Another proposed mechanism is the dissociation of AV-venom complexes, intensifying coagulopathy. If it recurs, retreatment with AV should be considered to prevent important bleeding.80 More knowledge about PK-PD parameters of AVs may support the understanding of this phenomenon.
WHO has recommended to establish public health strategies as an urgent measure to ensure the availability of AVs both safe, effective and affordable, especially for developing countries as well as to improve the regulatory control over the manufacture, import and sale.1 Therefore, it is necessary the implementation of PV in the emergency unit and ICU for achieve this objective.
Due to limited clinical information of the therapy with AVs, complementary tools of surveillance applied by hospital PV unit (HPU) are needed to ensure its quality, effectiveness, and safety post-marketing because they will have a crucial role since admission, during the intervention, transfer emergency to ICU, until discharge and post-ambulatory surveillance. Crisis setting within emergencies as well as in ICU exposes the patient to numerous medications, procedures and health care providers; therefore, an accurate and multidisciplinary intervention is necessary because there is a high risk of experiencing ADR, other drug-related problems, and making medication errors.81,82
The reported benefits of the intervention of HPU inside emergencies and in ICU result on improvements over safety culture, identification and classification of ADR, frequencies, decrease of costs related to ADR, among others.83-85
Since these areas are critical, HPU will provide support by means of tools and strategies to ensure quality of medication with the purpose of assessing the efficacy and safety of AVs in real practice.
Methods for the traceability before infusion
The technologies for traceability of a drug may ensure an infusion free from manufacturing errors.86 Indeed, the problem of counterfeit and substandard drugs is worrying.
In particular, WHO defines “substandard drugs” to those genuine medicines (with sanitary registry or good manufacturing practices) which have failed to pass the quality measurements and standards set for them.87 They are also known as “out of specification” and are often manufactured into emerging countries by unqualified personnel88 and the AVs could contain unknown impurities and bacteria (it release pyrogens).
Nonetheless, substandard AVs may be difficult to detect in the clinical practice because they seem identical to the previously approved product by regulatory agency and could not cause an ADR. However, they often would fail to properly treat the disease or condition they were intended to, and may lead to serious health consequences, including death. Fortunately, some technological innovations have been introduced in the manufacturing process for improving AVs quality,89 onsite PV systems, and product authentication systems.90
Monitoring during infusion protocol
ADR have been reported during the infusion protocol, especially during the first hours after administration. Syndrome of release of cytokines, hypersensitivity reactions, immunogenicity and delayed reactions may be present during the infusion.68-70
Hence, every hospital previously must set the guidelines for preparation of sterile and non-sterile mixtures. For example, Mexico has an Official Standard called NOM-249-SSA1-2010 about preparations (Sterile mixtures: nutritional, medicinal, infrastructure for their preparation) and current supplementary guidelines of pharmacopoeia for the good practices over the preparation of sterile and non-sterile mixtures.91 Moreover, the legal framework is based both on articles 198 (fraction IV), 258, 259, 261, 368, 369, 370 and 371 from Health General Law in Mexico,92 on articles 8, 99, 100, 109, 112 and 163 from Regulation of Health Products.93 The purpose of the guidelines is ensuring the identity, purity, concentration, potency, harmlessness, and safety of intravenous drugs, such as AVs.
Interestingly, it has been reported the use of transfer summaries “emergencies →ICU” as an effective tool to increase ADR detection and medication errors as complementary methods to the existing pharmaceutical services (i.e. medication prescription reviewed for appropriateness and its reconciliation).94-98 It may be useful for PV of AVs during and after infusion protocol.
Pharmacovigilance in toxicological emergencies
Emergency unit is aimed to receive patients in critical situations, with or without imminent risk of death. Unfortunately, there is not published data about the specific patterns of suspected ADR or other drug-related problems. However, indirect data has been reported a total of 6-10% of admissions by suspected ADR in emergencies.81 Therefore, an appropriate pharmaceutical intervention is absolutely need because the medication protocols are most complex compared to others medical areas. For example, a study reported the high prevalence (61%) of potential drug-drug interactions in patients admitted to emergency unit from a tertiary care hospital in Mexico.99
Similarly, a current study shows the effectiveness of a pharmaceutical intervention and it is addressed to physicians working in emergency and coordinated by a pharmacist in a Mexican pediatric hospital of tertiary care on the ADR identification and reporting. Here, >97% of ADR were correctly identified before and after the intervention, but only <7-42% of ADR were reported before and after the inter vention, respectively. The researchers conclude that emergency physicians identify a suspected ADR, but fail to report them. However, this study reported a significant increase over the number of reports of suspected ADR.100 This is an opportunity to add a PV system particularly in toxicological emergencies as a strategy to additionally guarantee the welfare of patient.
On the other hand, if patients have a life-threatening medical condition they will require constant monitoring of vital signs and other medical parameters in ICU. Here, the hospital attention is different from the emergency unit and a transfer to another ward is required. This moment is critical for a suitable PV of AVs. Notwithstanding, the clinical setting of the PV in ICU is different from emergencies. For example, an observational study reported a low incidence of ADR related to drug-drug interactions (DDI) in ICU due to strict control of this medical area.101 Likewise, there is a higher incidence of suspected ADR in ambulatory patients compared with the hospitalized patients;83 while a total of 7-15% of suspected ADR cases contributes significantly to ICU admissions.82,101 Thus, this evidence would support, at least, the hypothesis of a higher risk of ADR of AVs in the emergency units, mainly due to several complex circumstances of medical admission.
Notification of suspected ADR and causality assessment
The suspected ADR must be notified and subsequently evaluated for the causality assignment through algorithms. Indeed, the algorithms in PV are used to assess the causality of suspected ADR through flowcharts or assessment questions.
Currently, there is not a universally accepted method for assessing them, but according to the causality assessment, provided by WHO and UMC,102 there are probabilistic categories called —Certain, Probable/Likely, Possible, Unlikely, Conditional/Unclassified, Non-assessable/Unclassifiable—. Hence, there are several algorithms, but the best known is Naranjo’s algorithm. Table 3 shows the comparison among them, highlighting the advantages and limitations in real practice.
On other hand, the similarity among the three algorithms (Naranjo, Jones and Begaud) has been assessed in the diagnosis of drug hypersensitivity; the ADR most frequent with the treatment of AV. The authors indicated dissimilarity between the algorithms, but they agree with Jones’ and Naranjo’s methods.104 In addition, the results were similar when evaluating ADR in the ICU using Naranjo’s, Kramer’s and Jones’ algorithms. Authors suggested an instrument selection with any of the three instruments will be reasonable but there is lower variability with the Kramer method; however, it is a very long appraisal which will be little practical both at the clinical setting and at toxicological emergencies.103
| Table 3. Advantages and limitations of causality assessment tools* | ||
|---|---|---|
| Method of causality appraisal | Advantages against other methods | Limitations against other methods |
| Karch and Lasagna’s algorithm | None | Reliability and validity have not been well established. |
| Kramer’s scale | None | Employ exhaustive flowcharts, excessively intricate and protracted for realistic application. |
| Naranjo’s algorithm | Simple and brief, most extensively used | Dependability and validity not confirmed in children or other risk groups. Stronger dependence on the meaning given to the “possible” category. It does not offer sufficient information for making decisions. |
| Begaud’s algorithm | None | Its application requires 3-stage flowchart, not protracted, but unable to employ all feature characteristically utilized in ADR appraisal. |
| Jones’s algorithm | Shorter and quicker to detect ADR | It cannot identify actual cause |
| Global introspection | Most common approach, more sensitive | Inter- and intra-rater contradiction; subjectivity and imprecision; poor reproducibility because it is mainly based on expert clinical judgements. |
| *Based in the table of Marante KB (2018)103 | ||
Proposal for a modified Naranjo’s algorithm
A modification of the Naranjo’s algorithm is proposed here because it would be a fast and feasible algorithm used for cases of suspected ADR with AVs. Moreover, the modification is necessary because 1) the effectiveness and safety profile between the AVs worldwide has not been widely shown; 2) there is a complex setting between the admission and transfer of a patient; 3) frequency and type of suspected ADR with AVs, and especially 4) because the signs and symptoms of the suspected ADR often may be confused with those related to the natural history from envenoming or the ADR of the other conventional drugs used of infusion therapy (i.e. prazosin). Thus, a modified Naranjo’s algorithm may help to increase the clinical acuity for distinguish them.
Although Naranjo’s algorithm does not provide high sensitivity and specificity by itself over particular cases, it is possible that a change may be useful to a “quicker check list” in toxicological emergencies or an initial appraisal of suspected ADR.
The modification consists of replacing questions 6 and 7 because they would not be related to clinical setting of AVs and they may be confusing factors. These replaced questions are related to the clinical investigation and therapeutic monitoring by laboratory but not to the real clinical practice of treatment.
Frequently, when the causality of a suspected ADR is researched in the PV practice with AVs, these questions (6 and 7) are answered as “unknown”. This would affect the scale as well as the sensitivity and specificity of causality grade by high likelihood to be classified as “possible”. In order to decrease it, this proposal consist of replacing these questions for others related to clinical questions at issue of AVs for decision making in toxicological emergencies. It will not modify the scale because they have the same logic. Therewith, it would be possible an increase in the sensitivity and specificity about causality grade of suspected ADR.
Table 4 shows the modified Naranjo’s algorithm proposed here. Notwithstanding, a clinical study will be need to confirm the validity of this proposal as future perspectives.
| Table 4. Modified Naranjo’s algorithm: ADR related to treatment with AV. | |||
|---|---|---|---|
| Question | Yes | No | Unknown |
| 1. Are there previous conclusive reports on this reaction? | +1 | 0 | 0 |
| 2. Did the adverse event appear after the suspected drug was administered? | +2 | -1 | 0 |
| 3. Did the adverse event improve when the drug was discontinued or a specific antagonist was administered? | +1 | 0 | 0 |
| 4. Did the adverse event reappear when the drug was re-administered? | +2 | -1 | 0 |
| 5. Are there alternative causes that could on their own have caused the reaction? | -1 | +2 | 0 |
| *6. Were the doses or number of vials according with the instructions from manufacturer? | -1 | +1 | 0 |
| **7. Was the estimated time ≤24 h between the medical attention and the AV administration? | +1 | 0 | 0 |
| 8. Was the reaction more severe when the dose was increased or less severe when the dose was decreased? | +1 | 0 | 0 |
| 9. Did the patient have a similar reaction to the same or similar drugs in any previous exposure? | +1 | 0 | 0 |
| 10. Was the adverse event confirmed by any objective evidence? | +1 | 0 | 0 |
| ***Total | |||
| *Original question: Did reaction reappear when a placebo was given? Rational of change: Placebo would be unacceptable and the term is focusing on clinical research not on the medical practice. **Original question: Was the drug detected in blood or other fluids in concentrations known to be toxic? Rational of change: Therapeutic monitoring is not mandatory or routine in the treatment of envenoming. ***Total Score (ADR Probability Scale): ≥9 (definite), 5-8 (probable), 1-4 (possible), ≤0 (doubtful) | |||
Quality of PV report
It is widely dependent on the quality of the clinical file; thus, anecdotal great efforts have been established for improving both of them. According to the Mexican Official Standard NOM 220-SSA1-2016 dealing with PV, there is certain information grade based on the data received in the notifications as follows: 1) Grade 0, it includes an identifiable patient, at least one ADR or some other safety problems related to the use of medications and vaccines, suspect medication or vaccine and data from the notifier; 2) Grade 1, idem + it includes start dates of the ADR, date of beginning of treatment and date of treatment end (day, month and year); 3) Grade 2, idem + it includes generic denomination, distinctive name, dosage, route of administration, reason for prescription, consequence of the event, important data of the clinical records, batch number, and manufacturer’s name; 4) Grade 3, idem + it includes the result of the re-administration of the medication or vaccine.
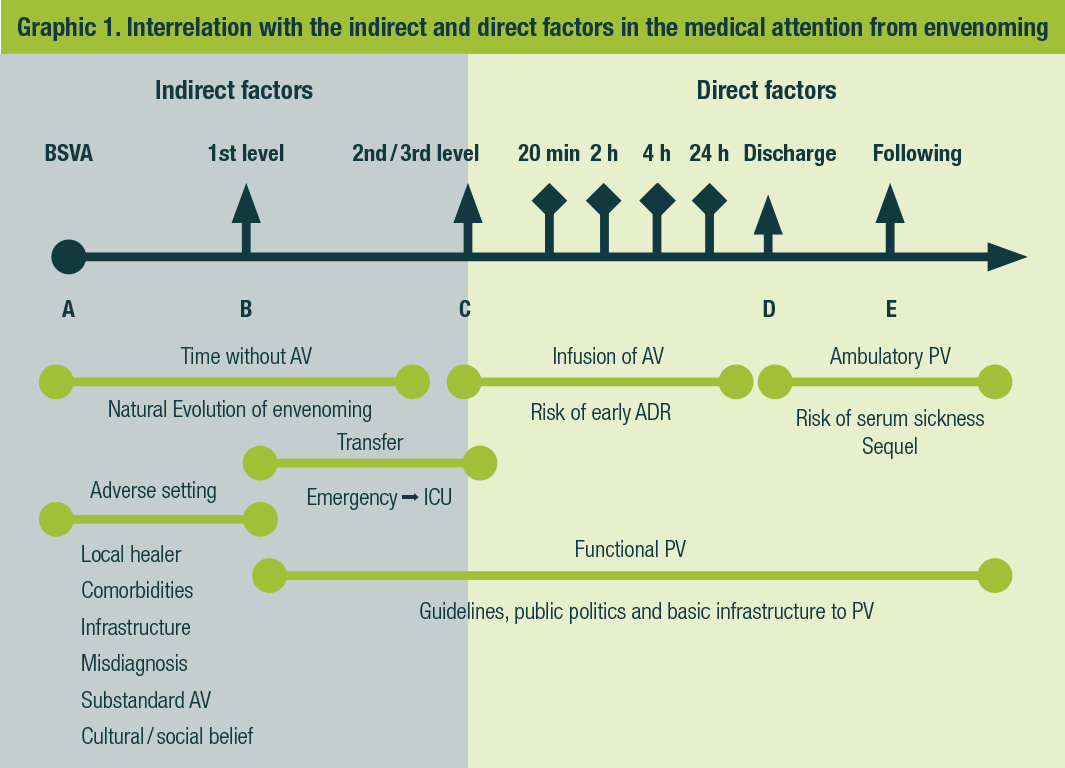
The goal will be reported to the sanitary authority, as well as the ADR associated to AV with a high grade of information (between 2-3) and the higher causality (understanding previously the clinical setting of AV treatment) to support the decision making and avoidability. Since the PV report quality would be influenced by indirect and direct factors of medical attention, it is necessary to know the envenoming setting as a whole. Graphic 1 shows the interrelation with those factors.
Follow-up after discharge
The PV in the envenoming ambulatory care is a challenge because the patients with favorable prognosis and subsequent discharge have, at least, a sequel or signs or symptoms of serum sickness (until 5-15 days ago), but they do not often return to the hospital. Thus, it is recommended that HPU gathers data from patients and asks for the informed consent to be subsequently contacted by a health professional with the purpose of carrying out the follow-up after a successfully solved the envenoming crisis. Besides, several studies described the electronic and mobile resources for medication safety and PV to enhance patient safety in the ambulatory care setting;105 thus, they may be implemented by HPU with a few respective modifications.
There are other AV-related factors which indirectly may affect the effectiveness and safety of the treatment; for instance, tourniquets are generally not recommended for snakebites due to high risk of fatality. Likewise, traditional manipulations like local incision and suction are not recommended.15 If the patient arrived with a tourniquet already placed by a family member or rescuer before being admitted to emergencies, do not to remove (as some experts have recommended) the pressure until prepared to manage acute toxicity.106,107
Commonly, the patients living in rural areas go to the local healer —Curandero— before going to the first contact physician. Then, the patients are submitted to unknown procedures such as consuming herbal remedies which could interact with other drugs both in emergencies and ICU.108,109 Moreover, a long delay because of a slow transportation and a lack of available transport are some factors limiting the timely access to AV, and accordingly, to a favorable prognosis.2,37 Physicians and HPU must record these variables in the medical record because they are associated to therapeutic fail and other complications (see Graphic 1).
Envenoming by BSVA is a toxicological emergency whereby the venom is injected to a patient through the bite or sting of a venomous animal (i.e. snake, spider and scorpion) or ingestion of toxins produced by animals or plants. It is a miscellaneous medical situation; but it is generally damaged the hematological, renal, cardiovascular and neuronal system since a mild grade until a human life-threatening. Here, the drug management and control among “emergency → ICU” is critical to a safe therapeutic response.
AVs are biological therapies indicated in the treatment of envenoming by BSVA. However, the access to the AVs is a neglected public health problem, especially in emergent countries as Mexico apparently for the low prevalence of registered accidents by BSVA. Moreover, the lack of robust clinical trials, medical consensus and drug quality problems (i.e. counterfeit) may trigger the non-safety and inappropriate use of AV. All they are PV-related factors of AVs. Therefore, the implementation of hospital strategies for the strengthening of safety patient are needed. Consequently, the PV is a hospital activity focused on improving the quality, effectiveness, and safety of AVs in cases of toxicological emergencies.
In particular, the AVs have pharmacological parameters different from conventional drugs and the patterns of ADR are associated with type 1 hypersensitivity reactions during infusion protocol; the natural evolution of envenoming could be confused with a suspected ADR. Therefore, an adequacy in the algorithms for the assessment of causality of the suspected ADR is suitable. Therefore, a modified Naranjo’s algorithm has been proposed to increase the sensitivity and specificity of the assessment of causality of suspected ADR within clinical setting of toxicological emergencies. However, more prospective studies are needed to validate this hypothesis.
As perspective, the activities of the PV and the hospital pharmaceutical services are governed under international standards; they must not be an “independent check list” but supplementary activities within an integral surveillance system of medication along with the government programs directed by local authorities. Unfortunately, these standards as well as the PV are poorly known in tropical regions such as Latin America, Middle East, Africa and Asia because there are sociocultural and infrastructural barriers whereby delay or undernotify the PV report or other drug-related problems.
In conclusion, the PV must be established as a routine activity within an integral process of the medical attention of envenoming by BSVA to guarantee a suitable and safe medication.
We thank Inosan Biopharma España S.A. for the editorial support of this document by means of Vesalio Difusión Médica S.A. de C.V.
A.E. has been a scientific consultant and a lecturer for Eli Lilly, Sanofi Aventis, Ferring, Janssen, Abbvie, Novartis, and Pfizer.
G.M. declares no conflict of interest. R.S. and N.V. work for Inosan Biopharma S.A.
| 1. | World Health Organization. WHO | WHO Guidelines for the Production, Control and Regulation of Snake Antivenom Immunoglobulins. WHO [Internet]. 2018 [cited 2018 Aug 29]; Available from: http://www.who.int/bloodproducts/snake_antivenoms/snakeantivenomguide/en/ |
| 2. | Iliyasu G, Tiamiyu AB, Daiyab FM, Tambuwal SH, Habib ZG, Habib AG. Effect of distance and delay in access to care on outcome of snakebite in rural north-eastern Nigeria. Rural Remote Health [Internet]. 2015 [cited 2018 Nov 8];15(4):3496. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26590373 |
| 3. | Sheehan WJ, Gaffin JM, Peden DB, Bush RK, Phipatanakul W. Advances in environmental and occupational disorders in 2016. J Allergy Clin Immunol [Internet]. 2017 Dec [cited 2018 Nov 8];140(6):1683–92. Available from: https://linkinghub.elsevier.com/retrieve/pii/S009167491731655X |
| 4. | Giezen TJ, Mantel-Teeuwisse AK, Leufkens HGM. Pharmacovigilance of biopharmaceuticals: challenges remain. Drug Saf [Internet]. 2009 Oct [cited 2018 Nov 8];32(10):811–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19722725 |
| 5. | NORMA Oficial Mexicana NOM-220- SSA1-2016, instalación y operación de la farmacovigilancia [Internet]. 2016 [cited 2018 Aug 29]. Available from: http://www.dof.gob.mx/nota_detalle.php?codigo=5490830&fecha=19/07/2017 |
| 6. | International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. Pharmacovigilance Planning : ICH [Internet]. [cited 2018 Jul 10]. Available from: https://www.ich.org/fileadmin/Public_Web_Site/ICH_ Products/Guidelines/Efficacy/E2E/Step4/E2E_Guideline.pdf |
| 7. | Castro-Pastrana LI, Carleton BC. Improving pediatric drug safety: need for more efficient clinical translation of pharmacovigilance knowledge. J Popul Ther Clin Pharmacol [Internet]. 2011 [cited 2018 Nov 8];18:e76-88. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21467599 |
| 8. | Vázquez-Alvarez AO, Brennan-Bourdon LM, Rincón-Sánchez AR, Islas-Carbajal MC, Huerta-Olvera SG. Improved drug safety through intensive pharmacovigilance in hospitalized pediatric patients. BMC Pharmacol Toxicol [Internet]. 2017 Dec 8 [cited 2018 Nov 8];18(1):79. Available from: https://bmcpharmacoltoxicol.biomedcentral.com/articles/10.1186/s40360-017-0186-x |
| 9. | White J. Venomous animals: clinical toxinology. EXS [Internet]. 2010 [cited 2018 Nov 8];100:233–91. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20358686 |
| 10. | Krau SD. Bites and stings: epidemiology and treatment. Crit Care Nurs Clin North Am [Internet]. 2013 Jun [cited 2018 Nov 8];25(2):143–50. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0899588513000166 |
| 11. | Gutiérrez JM. Reducing the impact of snakebite envenoming in Latin America and the Caribbean: achievements and challenges ahead. Trans R Soc Trop Med Hyg [Internet]. 2014 Sep [cited 2018 Nov 8];108(9):530–7. Available from: https://academic.oup.com/trstmh/article-lookup/doi/10.1093/trstmh/tru102 |
| 12. | Bannister JB. Regulating Rare Disease: Safely Facilitating Access to Orphan Drugs. Fordham law Rev [Internet]. 2018 Mar [cited 2018 Nov 8];86(4):1889–921. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29993206 |
| 13. | Dirección General de Epidemiología. Anuarios de Morbilidad 1984 - 2017 | Secretaría de Salud | Gobierno | gob.mx [Internet]. [cited 2018 Aug 10]. Available from: https://www. gob.mx/salud/acciones-y-programas/anuarios-de-morbilidad-1984-2017 |
| 14. | Santos MS V, Silva CGL, Neto BS, Grangeiro Júnior CRP, Lopes VHG, Teixeira Júnior AG, et al. Clinical and Epidemiological Aspects of Scorpionism in the World: A Systematic Review. Wilderness Environ Med [Internet]. 2016 Dec [cited 2018 Nov 8];27(4):504–18. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1080603216302149 |
| 15. | Chippaux J-P, Goyffon M. Epidemiology of scorpionism: A global appraisal. Acta Trop [Internet]. 2008 Aug [cited 2018 Nov 8];107(2):71–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18579104 |
| 16. | Gummin DD, Mowry JB, Spyker DA, Brooks DE, Fraser MO, Banner W. 2016 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 34th Annual Report. Clin Toxicol (Phila) [Internet]. 2017 Dec 26 [cited 2018 Nov 8];55(10):1072–252. Available from: https://www.tandfonline.com/doi/full/10.1080/1556 3650.2017.1388087 |
| 17. | Langley R, Mack K, Haileyesus T, Proescholdbell S, Annest JL. National estimates of noncanine bite and sting injuries treated in US Hospital Emergency Departments, 2001- 2010. Wilderness Environ Med [Internet]. 2014 Mar [cited 2018 Nov 8];25(1):14–23. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1080603213002482 |
| 18. | Isbister GK, Fan HW. Spider bite. Lancet [Internet]. 2011 Dec [cited 2018 Nov 8];378(9808):2039–47. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0140673610622301 |
| 19. | Diaz JH. The global epidemiology, syndromic classification, management, and prevention of spider bites. Am J Trop Med Hyg [Internet]. 2004 Aug [cited 2018 Nov 8];71(2):239–50. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15306718 |
| 20. | Gutiérrez JM, Calvete JJ, Habib AG, Harrison RA, Williams DJ, Warrell DA. Snakebite envenoming. Nat Rev Dis Prim [Internet]. 2017 Sep 14 [cited 2018 Nov 9];3:17063. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28905944 |
| 21. | Corbett B, Clark RF. North American Snake Envenomation. Emerg Med Clin North Am [Internet]. 2017 May [cited 2018 Nov 9];35(2):339–54. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0733862716301171 |
| 22. | Utkin YN. Animal venom studies: Current benefits and future developments. World J Biol Chem [Internet]. 2015 May 26 [cited 2018 Nov 8];6(2):28–33. Available from: http://www.wjgnet.com/1949-8454/full/v6/i2/28.htm |
| 23. | Kularatne SAM, Senanayake N. Venomous snake bites, scorpions, and spiders. Handb Clin Neurol [Internet]. 2014 [cited 2018 Nov 8];120:987–1001. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780702040870000668 |
| 24. | Del Brutto OH. Neurological effects of venomous bites and stings. In: Handbook of clinical neurology [Internet]. 2013 [cited 2018 Nov 9]. p. 349–68. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23829924 |
| 25. | Khattabi A, Soulaymani-Bencheikh R, Achour S, Salmi L-R, Scorpion Consensus Expert Group. Classification of clinical consequences of scorpion stings: consensus development. Trans R Soc Trop Med Hyg [Internet]. 2011 Jul [cited 2018 Nov 9];105(7):364–9. Available from: https://academic.oup.com/trstmh/article-lookup/doi/10.1016/j.trstmh.2011.03.007 |
| 26. | Shackleford R, Veillon D, Maxwell N, LaChance L, Jusino T, Cotelingam J, et al. The black widow spider bite: differential diagnosis, clinical manifestations, and treatment options. J La State Med Soc [Internet]. [cited 2018 Nov 9];167(2):74–8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25978056 |
| 27. | Haddad V, Cardoso JLC, Lupi O, Tyring SK. Tropical dermatology: Venomous arthropods and human skin: Part II. Diplopoda, Chilopoda, and Arachnida. J Am Acad Dermatol [Internet]. 2012 Sep [cited 2018 Nov 9];67(3):347.e1-9; quiz 355. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0190962212006226 |
| 28. | Hifumi T, Sakai A, Kondo Y, Yamamoto A, Morine N, Ato M, et al. Venomous snake bites: clinical diagnosis and treatment. J intensive care [Internet]. 2015 Dec 1 [cited 2018 Nov 9];3(1):16. Available from: http://www.jintensivecare.com/content/3/1/16 |
| 29. | Weinstein S, Dart R, Staples A, White J. Envenomations: an overview of clinical toxinology for the primary care physician. Am Fam Physician [Internet]. 2009 Oct 15 [cited 2018 Nov 9];80(8):793–802. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19835341 |
| 30. | Wijesinghe CA, Williams SS, Kasturiratne A, Dolawaththa N, Wimalaratne P, Wijewickrema B, et al. A Randomized Controlled Trial of a Brief Intervention for Delayed Psychological Effects in Snakebite Victims. Franco-Paredes C, editor. PLoS Negl Trop Dis [Internet]. 2015 Aug 11 [cited 2018 Nov 9];9(8):e0003989. Available from: https://dx.plos.org/10.1371/journal.pntd.0003989 |
| 31. | da Silva WD, Tambourgi D V. The humoral immune response induced by snake venom toxins. Inflamm Allergy Drug Targets [Internet]. 2011 Oct [cited 2018 Nov 9];10(5):343–57. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21824082 |
| 32. | León G, Sánchez L, Hernández A, Villalta M, Herrera M, Segura A, et al. Immune response towards snake venoms. Inflamm Allergy Drug Targets [Internet]. 2011 Oct [cited 2018 Nov 9];10(5):381–98. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21824081 |
| 33. | Bouhaouala-Zahar B, Ben Abderrazek R, Hmila I, Abidi N, Muyldermans S, El Ayeb M. Immunological aspects of scorpion toxins: current status and perspectives. Inflamm Allergy Drug Targets [Internet]. 2011 Oct [cited 2018 Nov 9];10(5):358–68. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21756244 |
| 34. | Klotz JH, Klotz SA, Pinnas JL. Animal bites and stings with anaphylactic potential. J Emerg Med [Internet]. 2009 Feb [cited 2018 Nov 9];36(2):148–56. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0736467907004167 |
| 35. | El-DeeK SEM, Sayed AA, Nassar AY, Mohey- Eldeen ZM, Eldeeb HM, Meki A-RMA. Role of some vasoactive mediators in scorpion envenomed children: Possible relation to envenoming outcome. Toxicon [Internet]. 2017 Mar 1 [cited 2018 Nov 9];127:77–84. Available from: https://linkinghub.elsevier.com/retrieve/pii/S004101011730017X |
| 36. | Hsu C-P, Chuang J-F, Hsu Y-P, Wang S-Y, Fu C-Y, Yuan K-C, et al. Predictors of the development of post-snakebite compartment syndrome. Scand J Trauma Resusc Emerg Med [Internet]. 2015 Nov 11 [cited 2018 Nov 9];23(1):97. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4642665/ |
| 37. | McKinney PE. Out-of-hospital and interhospital management of crotaline snakebite. Ann Emerg Med [Internet]. 2001 Feb [cited 2018 Nov 9];37(2):168–74. Available from: https://www.ncbi.nlm.nih.gov/pubmed/11174235 |
| 38. | Simpson ID. Snakebite management in India, the first few hours: a guide for primary care physicians. J Indian Med Assoc [Internet]. 2007 Jun [cited 2018 Nov 9];105(6):324, 326, 328 passim. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18232178 |
| 39. | Haddad Junior V, Amorim PCH de, Haddad Junior WT, Cardoso JLC. Venomous and poisonous arthropods: identification, clinical manifestations of envenomation, and treatments used in human injuries. Rev Soc Bras Med Trop [Internet]. 2015 Dec [cited 2018 Nov 9];48(6):650–7. Available from: http://www.scielo.br/scielo.php?script=sci_arttext& pid=S0037-86822015000600650&lng= en&tlng=en |
| 40. | Juckett G, Hancox JG. Venomous snakebites in the United States: management review and update. Am Fam Physician [Internet]. 2002 Apr 1 [cited 2018 Nov 9];65(7):1367–74. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11996419 |
| 41. | Johnston CI, Ryan NM, Page CB, Buckley NA, Brown SG, O’Leary MA, et al. The Australian Snakebite Project, 2005-2015 (ASP-20). Med J Aust [Internet]. 2017 Aug 7 [cited 2018 Nov 9];207(3):119–25. Available from: https://www.mja.com.au/journal/2017/207/3/australian-snakebite-project-2005-2015-asp-20 |
| 42. | Cupo P. Clinical update on scorpion envenoming. Rev Soc Bras Med Trop [Internet]. 2015 Dec [cited 2018 Nov 9];48(6):642–9. Available from: http://www.scielo.br/scielo.php?script=sci_arttext& pid=S0037-86822015000600642&lng= en&tlng=en |
| 43. | Chippaux J-P. Emerging options for the management of scorpion stings. Drug Des Devel Ther [Internet]. 2012 Jul [cited 2018 Nov 9];6:165–73. Available from: http://www.dovepress.com/emerging-options-for-themanagement-of-scorpion-stings-peer-reviewed-article-DDDT |
| 44. | Diaz JH, Leblanc KE. Common spider bites. Am Fam Physician [Internet]. 2007 Mar 15 [cited 2018 Nov 9];75(6):869–73. Available from: https://www.ncbi.nlm.nih.gov/pubmed/17390599 |
| 45. | Lavonas EJ, Ruha A-M, Banner W, Bebarta V, Bernstein JN, Bush SP, et al. Unified treatment algorithm for the management of crotaline snakebite in the United States: results of an evidence-informed consensus workshop. BMC Emerg Med [Internet]. 2011 Feb 3 [cited 2018 Nov 9];11(1):2. Available from: http://bmcemergmed.biomedcentral.com/articles/10.1186/1471-227X-11-2 |
| 46. | Naik BS. “Dry bite” in venomous snakes: A review. Toxicon [Internet]. 2017 Jul [cited 2018 Nov 9];133:63–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28456535 |
| 47. | Theakston RDG, Warrell DA, Griffiths E. Report of a WHO workshop on the standardization and control of antivenoms. Toxicon [Internet]. 2003 Apr [cited 2018 Nov 9];41(5):541–57. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12676433 |
| 48. | Gutiérrez JM, León G, Lomonte B. Pharmacokinetic- pharmacodynamic relationships of immunoglobulin therapy for envenomation. Clin Pharmacokinet [Internet]. 2003 [cited 2018 Nov 9];42(8):721–41. Available from: http://link.springer.com/10.2165/00003088-200342080-00002 |
| 49. | Lobo ED, Hansen RJ, Balthasar JP. Antibody pharmacokinetics and pharmacodynamics. J Pharm Sci [Internet]. 2004 Nov [cited 2018 Nov 9];93(11):2645–68. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0022354916316379 |
| 50. | Salud S de. Guía de práctica clínica. Prevención, diagnóstico, tratamiento y referencia de la intoxicación por picadura de alacrán [Internet]. 2015. Available from: http://www.cenetec-difusion.com/CMGPC/SS-148-08/ER.pdf |
| 51. | SALUD, SEDENA S. Guía de práctica clínica. Diagnóstico y tratamiento de mordeduras por arañas venenosas [Internet]. México; 2011. Available from: http://www.cenetec-difusion.com/CMGPC/SS-523-11/ER.pdf |
| 52. | SALUD, SEDENA S. Diagnóstico y tratamiento de las mordeduras de serpientes venenosas. pdf [Internet]. 2010. Available from: http://www.cenetec-difusion.com/CMGPC/SS-298-10/ER.pdf |
| 53. | de Roodt AR, Clement H, Dolab JA, Litwin S, Hajos SE, Boyer L, et al. Protein content of antivenoms and relationship with their immunochemical reactivity and neutralization assays. Clin Toxicol (Phila) [Internet]. 2014 Jul 18 [cited 2018 Nov 9];52(6):594–603. Available from: http://www.tandfonline.com/doi/full/10.3109/15563650.2014.925561 |
| 54. | Madrigal M, Pla D, Sanz L, Barboza E, Arroyo-Portilla C, Corrêa-Netto C, et al. Cross-reactivity, antivenomics, and neutralization of toxic activities of Lachesis venoms by polyspecific and monospecific antivenoms. Casewell NR, editor. PLoS Negl Trop Dis [Internet]. 2017 Aug 7 [cited 2018 Nov 9];11(8):e0005793. Available from: https://dx.plos.org/10.1371/journal.pntd.0005793 |
| 55. | Maria WS, Velarde DT, Alvarenga LM, Nguyen C, Villard S, Granier C, et al. Localization of epitopes in the toxins of Tityus serrulatus scorpions and neutralizing potential of therapeutic antivenoms. Toxicon [Internet]. 2005 Aug [cited 2018 Nov 9];46(2):210–7. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0041010105001352 |
| 56. | Buch DR, Souza FN, Meissner GO, Morgon AM, Gremski LH, Ferrer VP, et al. Brown spider (Loxosceles genus) venom toxins: Evaluation of biological conservation by immune cross-reactivity. Toxicon [Internet]. 2015 Dec 15 [cited 2018 Nov 9];108:154–66. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0041010115300982 |
| 57. | Mix E, Goertsches R, Zett UK. Immunoglobulins— Basic considerations. J Neurol [Internet]. 2006 Sep [cited 2018 Nov 9];253(S5):v9–17. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16998762 |
| 58. | Engels N, Wienands J. Memory control by the B cell antigen receptor. Immunol Rev [Internet]. 2018 May [cited 2018 Nov 9];283(1):150–60. Available from: http://doi.wiley.com/10.1111/imr.12651 |
| 59. | Wilde H, Thipkong P, Sitprija V, Chaiyabutr N. Heterologous antisera and antivenins are essential biologicals: perspectives on a worldwide crisis. Ann Intern Med [Internet]. 1996 Aug 1 [cited 2018 Nov 9];125(3):233–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8686982 |
| 60. | Laustsen AH, María Gutiérrez J, Knudsen C, Johansen KH, Bermúdez-Méndez E, Cerni FA, et al. Pros and cons of different therapeutic antibody formats for recombinant antivenom development. Toxicon [Internet]. 2018 May [cited 2018 Nov 9];146:151–75. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0041010118301144 |
| 61. | Bumbaca D, Boswell CA, Fielder PJ, Khawli LA. Physiochemical and biochemical factors influencing the pharmacokinetics of antibody therapeutics. AAPS J [Internet]. 2012 Sep 18 [cited 2018 Nov 9];14(3):554–8. Available from: http://www.springerlink.com/index/10.1208/s12248-012-9369-y |
| 62. | Sanhajariya S, Duffull SB, Isbister GK. Pharmacokinetics of Snake Venom. Toxins (Basel) [Internet]. 2018 Feb 7 [cited 2018 Nov 9];10(2):73. Available from: http://www.mdpi.com/2072-6651/10/2/73 |
| 63. | Krifi MN, Savin S, Debray M, Bon C, El Ayeb M, Choumet V. Pharmacokinetic studies of scorpion venom before and after antivenom immunotherapy. Toxicon [Internet]. 2005 Feb [cited 2018 Nov 9];45(2):187–98. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0041010104004374 |
| 64. | Ferl GZ, Theil F-P, Wong H. Physiologically based pharmacokinetic models of small molecules and therapeutic antibodies: a mini-review on fundamental concepts and applications. Biopharm Drug Dispos [Internet]. 2016 Mar [cited 2018 Nov 9];37(2):75–92. Available from: http://doi.wiley.com/10.1002/bdd.1994 |
| 65. | Xiao JJ. Pharmacokinetic models for FcRn-mediated IgG disposition. J Biomed Biotechnol [Internet]. 2012 [cited 2018 Nov 9];2012:282989. Available from: http://www.hindawi.com/journals/bmri/2012/282989/ |
| 66. | Diao L, Meibohm B. Pharmacokinetics and pharmacokinetic-pharmacodynamic correlations of therapeutic peptides. Clin Pharmacokinet [Internet]. 2013 Oct 30 [cited 2018 Nov 9];52(10):855–68. Available from: http://link.springer.com/10.1007/s40262-013-0079-0 |
| 67. | Lavonas EJ. Antivenoms for snakebite: design, function, and controversies. Curr Pharm Biotechnol [Internet]. 2012 Aug [cited 2018 Nov 9];13(10):1980–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22352733 |
| 68. | Deshpande RP, Motghare VM, Padwal SL, Pore RR, Bhamare CG, Deshmukh VS, et al. Adverse drug reaction profile of anti-snake venom in a rural tertiary care teaching hospital. J Young Pharm [Internet]. 2013 Jun [cited 2018 Nov 9];5(2):41–5. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24396245 |
| 69. | Isbister GK. Safety of i.v. administration of redback spider antivenom. Intern Med J [Internet]. 2007 Dec [cited 2018 Nov 9];37(12):820–2. Available from: http://doi.wiley.com/10.1111/j.1445-5994.2007.01513.x |
| 70. | Mong R, Ng VCH, Tse ML. Safety profile of snake antivenom (use) in Hong Kong - a review of 191 cases from 2008 to 2015. Clin Toxicol (Phila) [Internet]. 2017 Dec 26 [cited 2018 Nov 9];55(10):1066–71. Available from: https://www.tandfonline.com/doi/full/10.1080/15563650.2017.1334916 |
| 71. | de Silva HA, Ryan NM, de Silva HJ. Adverse reactions to snake antivenom, and their prevention and treatment. Br J Clin Pharmacol [Internet]. 2016 Mar [cited 2018 Nov 9];81(3):446–52. Available from: http://doi.wiley.com/10.1111/bcp.12739 |
| 72. | Morais V. Antivenom therapy: efficacy of premedication for the prevention of adverse reactions. J Venom Anim Toxins Incl Trop Dis [Internet]. 2018 Dec 28 [cited 2018 Nov 9];24(1):7. Available from: https://jvat.biomedcentral.com/articles/10.1186/s40409-018-0144-0 |
| 73. | León G, Herrera M, Segura Á, Villalta M, Vargas M, Gutiérrez JM. Pathogenic mechanisms underlying adverse reactions induced by intravenous administration of snake antivenoms. Toxicon [Internet]. 2013 Dec 15 [cited 2018 Nov 9];76:63–76. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0041010113003607 |
| 74. | Smith A, Manoli H, Jaw S, Frutoz K, Epstein AL, Khawli LA, et al. Unraveling the Effect of Immunogenicity on the PK/PD, Efficacy, and Safety of Therapeutic Proteins. J Immunol Res [Internet]. 2016 [cited 2018 Nov 9];2016:2342187. Available from: http://www.hindawi.com/journals/jir/2016/2342187/ |
| 75. | Bloem K, Hernández-Breijo B, Martínez-Feito A, Rispens T. Immunogenicity of Therapeutic Antibodies: Monitoring Antidrug Antibodies in a Clinical Context. Ther Drug Monit [Internet]. 2017 Aug [cited 2018 Nov 9];39(4):327–32. Available from: http://insights.ovid.com/crossref?an=00007691-201708000-00005 |
| 76. | Ryan NM, Downes MA, Isbister GK. Clinical features of serum sickness after Australian snake antivenom. Toxicon [Internet]. 2015 Dec 15 [cited 2018 Nov 9];108:181–3. Available from: https://linkinghub.elsevier.com/retrieve/pii/S004101011530115X |
| 77. | Durandy A, Kaveri S V, Kuijpers TW, Basta M, Miescher S, Ravetch J V, et al. Intravenous immunoglobulins--understanding properties and mechanisms. Clin Exp Immunol [Internet]. 2009 Dec [cited 2018 Nov 9];158 Suppl 1:2–13. Available from: http://doi.wiley.com/10.1111/j.1365-2249.2009.04022.x |
| 78. | Seifert SA, I Kirschner R, Martin N. Recurrent, persistent, or late, new-onset hematologic abnormalities in Crotaline snakebite. Clin Toxicol (Phila) [Internet]. 2011 Apr 12 [cited 2018 Nov 9];49(4):324–9. Available from: http://www.tandfonline.com/doi/full/10.3109/15563650.2011.566883 |
| 79. | Boyer L V, Seifert SA, Clark RF, McNally JT, Williams SR, Nordt SP, et al. Recurrent and persistent coagulopathy following pit viper envenomation. Arch Intern Med [Internet]. 1999 Apr 12 [cited 2018 Nov 9];159(7):706–10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10218750 |
| 80. | Seifert SA, Boyer L V, Dart RC, Porter RS, Sjostrom L. Relationship of venom effects to venom antigen and antivenom serum concentrations in a patient with Crotalus atrox envenomation treated with a Fab antivenom. Ann Emerg Med [Internet]. 1997 Jul [cited 2018 Nov 9];30(1):49–53. Available from: http://www.ncbi.nlm.nih.gov/pubmed/9209225 |
| 81. | Schurig AM, Böhme M, Just KS, Scholl C, Dormann H, Plank-Kiegele B, et al. Adverse Drug Reactions (ADR) and Emergencies. Dtsch Arztebl Int [Internet]. 2018 Apr 13 [cited 2018 Nov 9];115(15):251–8. Available from: https://www.aerzteblatt.de/10.3238/arztebl.2018.0251 |
| 82. | Benkirane RR, Abouqal R, R-Abouqal R, Haimeur CC, S Ech Cherif El Kettani SS, Azzouzi AA, et al. Incidence of adverse drug events and medication errors in intensive care units: a prospective multicenter study. J Patient Saf [Internet]. 2009 Mar [cited 2018 Nov 9];5(1):16–22. Available from: https://insights.ovid.com/crossref?an=01209203-200903000-00004 |
| 83. | Batel Marques F, Penedones A, Mendes D, Alves C. A systematic review of observational studies evaluating costs of adverse drug reactions. Clinicoecon Outcomes Res [Internet]. 2016 Aug [cited 2018 Nov 9];8:413–26. Available from: https://www.ncbi.nlm.nih.gov/pubmed/27601925 |
| 84. | Schmiedl S, Rottenkolber M, Szymanski J, Drewelow B, Siegmund W, Hippius M, et al. Preventable ADRs leading to hospitalization — results of a long-term prospective safety study with 6,427 ADR cases focusing on elderly patients. Expert Opin Drug Saf [Internet]. 2018 Feb 19 [cited 2018 Nov 12];17(2):125–37. Available from: https://www.ncbi.nlm.nih.gov/pubmed/29258401 |
| 85. | Heavner JJ, Siner JM. Adverse Event Reporting and Quality Improvement in the Intensive Care Unit. Clin Chest Med [Internet]. 2015 Sep [cited 2018 Nov 12];36(3):461–7. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0272523115000660 |
| 86. | Ludasi K, Sovány T, Laczkovich O, Hopp B, Smausz T, Regdon G. Unique laser coding technology to fight falsified medicines. Eur J Pharm Sci [Internet]. 2018 Oct 15 [cited 2018 Nov 12];123:1–9. Available from: https://linkinghub.elsevier.com/retrieve/pii/S092809871830321X |
| 87. | Trippe ZA, Brendani B, Meier C, Lewis D. Identification of Substandard Medicines via Disproportionality Analysis of Individual Case Safety Reports. Drug Saf [Internet]. 2017 Apr 28 [cited 2018 Nov 12];40(4):293–303. Available from: https://ideas.repec.org/a/spr/drugsa/v40y2017i4d10.1007_s40264-016-0499-5.html |
| 88. | Almuzaini T, Choonara I, Sammons H. Substandard and counterfeit medicines: a systematic review of the literature. BMJ Open [Internet]. 2013 Aug 17 [cited 2018 Nov 12];3(8):e002923. Available from: http://bmjopen.bmj.com/lookup/doi/10.1136/bmjopen-2013-002923 |
| 89. | León G, Vargas M, Segura Á, Herrera M, Villalta M, Sánchez A, et al. Current technology for the industrial manufacture of snake antivenoms. Toxicon [Internet]. 2018 Sep 1 [cited 2018 Nov 12];151:63–73. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0041010118302952 |
| 90. | Fadlallah R, El-Jardali F, Annan F, Azzam H, Akl EA. Strategies and Systems-Level Interventions to Combat or Prevent Drug Counterfeiting: A Systematic Review of Evidence Beyond Effectiveness. Pharmaceut Med [Internet]. 2016 Oct 19 [cited 2018 Nov 12];30(5):263–76. Available from: http://link.springer.com/10.1007/s40290-016-0156-4 |
| 91. | DOF - Diario Oficial de la Federación. Norma Oficial Mexicana NOM-249-SSA1-2010, Mezclas estériles: nutricionales y medicamentosas, e instalaciones para su preparación [Internet]. 2011 [cited 2018 Aug 17]. Available from: http://dof.gob.mx/nota_detalle.php?codigo=5180629&fecha=04/03/2011 |
| 92. | Ley General de Salud. 2005;1–135. Available from: http://www.salud.gob.mx/cnts/pdfs/LEY_GENERAL_DE_SALUD.pdf |
| 93. | REGLAMENTO de Insumos para la Salud [Internet]. [cited 2018 Aug 17]. Available from: https://legislacion.vlex.com.mx/vid/reglamento-insumos-salud-387556665 |
| 94. | Hohl CM, Small SS, Peddie D, Badke K, Bailey C, Balka E. Why Clinicians Don’t Report Adverse Drug Events: Qualitative Study. JMIR public Heal Surveill [Internet]. 2018 Feb 27 [cited 2018 Nov 12];4(1):e21. Available from: http://publichealth.jmir.org/2018/1/e21/ |
| 95. | Anthes AM, Harinstein LM, Smithburger PL, Seybert AL, Kane-Gill SL. Improving adverse drug event detection in critically ill patients through screening intensive care unit transfer summaries. Pharmacoepidemiol Drug Saf [Internet]. 2013 May [cited 2018 Nov 12];22(5):510–6. Available from: https://www.ncbi.nlm.nih.gov/pubmed/23440931 |
| 96. | Zuppa AF, Adamson PC, Mondick JT, Davis LA, Maka DA, Narayan M, et al. Drug utilization in the pediatric intensive care unit: monitoring prescribing trends and establishing prioritization of pharmacotherapeutic evaluation of critically ill children. J Clin Pharmacol [Internet]. 2005 Nov [cited 2018 Nov 12];45(11):1305–12. Available from: http://doi.wiley.com/10.1177/0091270005280966 |
| 97. | Salud S de. Suplemento para establecimientos dedicados a la venta y suministro de medicamentos y demás insumos para la salud. [Internet]. 2018 [cited 2019 Mar 21]. Available from: https://www.farmacopea.org.mx/publicaciones-detalle.php?m=3&pid=4 |
| 98. | General CDS. Modelo De Seguridad Del Paciente del SiNaCEAM. 2018. 1-318 p. |
| 99. | Morales-Ríos O, Jasso-Gutiérrez L, Reyes-López A, Garduño-Espinosa J, Muñoz-Hernández O. Potential drug-drug interactions and their risk factors in pediatric patients admitted to the emergency department of a tertiary care hospital in Mexico. Eberini I, editor. PLoS One [Internet]. 2018 Jan 5 [cited 2018 Nov 12];13(1):e0190882. Available from: https://dx.plos.org/10.1371/journal.pone.0190882 |
| 100. | Ríos OM, Gutiérrez LJ, Talavera JO, Téllez-Rojo MM, López VO, Espinosa JG, et al. A comprehensive intervention for adverse drug reactions identification and reporting in a Pediatric Emergency Department. Int J Clin Pharm [Internet]. 2016 Feb 24 [cited 2018 Nov 12];38(1):80–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26499501 |
| 101. | Reis AMM, Cassiani SHDB. Adverse drug events in an intensive care unit of a university hospital. Eur J Clin Pharmacol [Internet]. 2011 Jun 19 [cited 2018 Nov 12];67(6):625–32. Available from: http://link.springer.com/10.1007/s00228-010-0987-y |
| 102. | Organization WH, Oreagba IA, Usman SO, Olayemi SO, Oshikoya KA, Opanuga O, et al. The use of the WHO-UMC system for standardized case causality assessment. Uppsala Uppsala Monit Cent [Internet]. 2014;48(3):194–203. Available from: http://www.who.int/medicines/areas/quality_safety/safety_efficacy/WHOcausality_assessment.pdf |
| 103. | Marante KB. The Challenges of Adverse Drug Reaction Evaluation. J Pharmacovigil [Internet]. 2018 [cited 2018 Nov 12];06(03). Available from: https://www.omicsonline.org/open-access/the-challenges-of-adverse-drug-reaction-evaluation-2329-6887-1000260-102439.html |
| 104. | Benahmed S, Picot MC, Hillaire-Buys D, Blayac JP, Dujols P, Demoly P. Comparison of pharmacovigilance algorithms in drug hypersensitivity reactions. Eur J Clin Pharmacol [Internet]. 2005 Aug 15 [cited 2018 Nov 12];61(7):537–41. Available from: http://link.springer.com/10.1007/s00228-005-0962-1 |
| 105. | Haas JS, Iyer A, Orav EJ, Schiff GD, Bates DW. Participation in an ambulatory e-pharmacovigilance system. Pharmacoepidemiol Drug Saf [Internet]. 2010 Sep [cited 2018 Nov 12];19(9):961–9. Available from: http://doi.wiley.com/10.1002/pds.2006 |
| 106. | Bush SP, Kinlaw SB. Management of a Pediatric Snake Envenomation After Presentation With a Tight Tourniquet. Wilderness Environ Med [Internet]. 2015 Sep [cited 2018 Nov 12];26(3):355–8. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1080603215000745 |
| 107. | García-López R, Villegas A, Pacheco-Coronel N, Gómez-Álvarez G. Traditional use and perception of snakes by the Nahuas from Cuetzalan del Progreso, Puebla, Mexico. J Ethnobiol Ethnomed [Internet]. 2017 Jan 21 [cited 2018 Nov 12];13(1):6. Available from: http://ethnobiomed.biomedcentral.com/articles/10.1186/s13002-016-0134-7 |
| 108. | Félix-Silva J, Silva-Junior AA, Zucolotto SM, Fernandes-Pedrosa M de F. Medicinal Plants for the Treatment of Local Tissue Damage Induced by Snake Venoms: An Overview from Traditional Use to Pharmacological Evidence. Evid Based Complement Alternat Med [Internet]. 2017 [cited 2018 Nov 12];2017:5748256. Available from: https://www.hindawi.com/journals/ecam/2017/5748256/ |
| 109. | Gutiérrez JM, Escalante T, Hernández R, Gastaldello S, Saravia-Otten P, Rucavado A. Why is Skeletal Muscle Regeneration Impaired after Myonecrosis Induced by Viperid Snake Venoms? Toxins (Basel) [Internet]. 2018 May 1 [cited 2018 Nov 12];10(5):182. Available from: http://www.mdpi.com/2072-6651/10/5/182 |
All Rights Reserved® 2019
Latin American Journal of Clinical Sciences and Medical Technology,Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.
All Rights Reserved® 2019
Publicación contínua • Editor responsable: Gilberto Castañeda Hernández. • Reserva de Derechos al Uso Exclusivo: 04-2019-062013242000-203; ISSN: 2683-2291; ambos otorgados por el Instituto Nacional del Derecho de Autor. • Responsable de la última actualización de este número, Web Master Hunahpú Velázquez Martínez,
Calle Profesor Miguel Serrano #8, Col. Del Valle, Alcaldía Benito Juárez, CP 03100, Ciudad de México, México. Número telefónico: 55 5405 1396 • Fecha de última modificación, 28 de agosto de 2024.